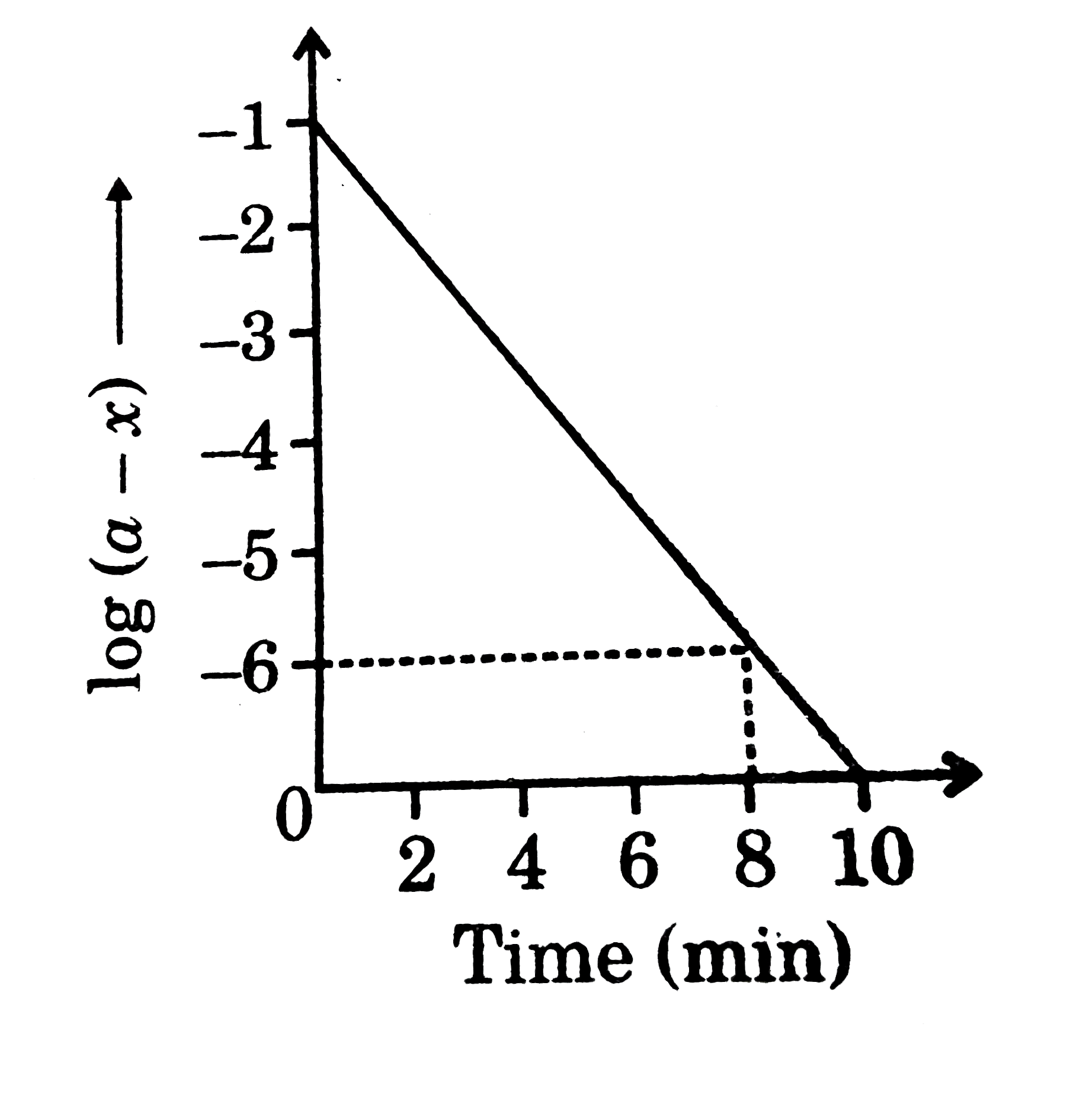
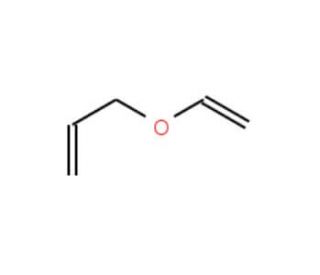
Reacting system geometries for the Claisen rearrangement of allyl vinyl... | Download Scientific Diagram

Activation of ether functionality of allyl vinyl ethers by chiral bis(organoaluminum) Lewis acids: application to asymmetric Claisen rearrangement - ScienceDirect
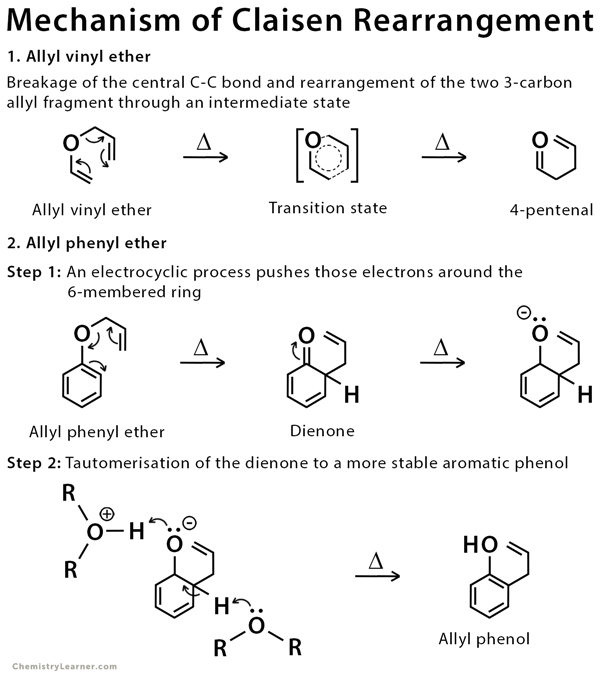
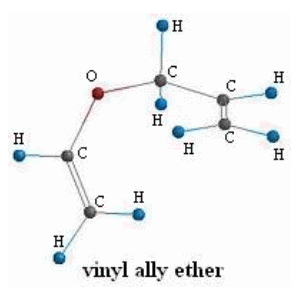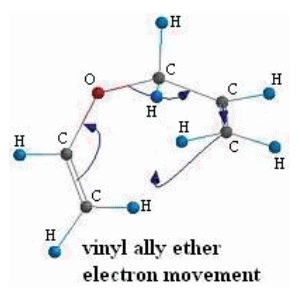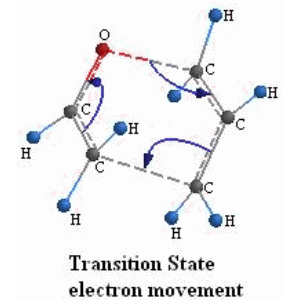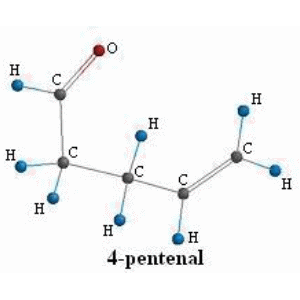
![PDF] Synthesis of alkenes: claisen rearrangement of allyl vinyl ethers, part i; mechanistic views; the organic chemistry notebook series, a didactical approach, N2 9 | Semantic Scholar PDF] Synthesis of alkenes: claisen rearrangement of allyl vinyl ethers, part i; mechanistic views; the organic chemistry notebook series, a didactical approach, N2 9 | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f7fadaa27890038feda7ad1d429351688b9cd998/5-Figure4-1.png)
PDF] Synthesis of alkenes: claisen rearrangement of allyl vinyl ethers, part i; mechanistic views; the organic chemistry notebook series, a didactical approach, N2 9 | Semantic Scholar
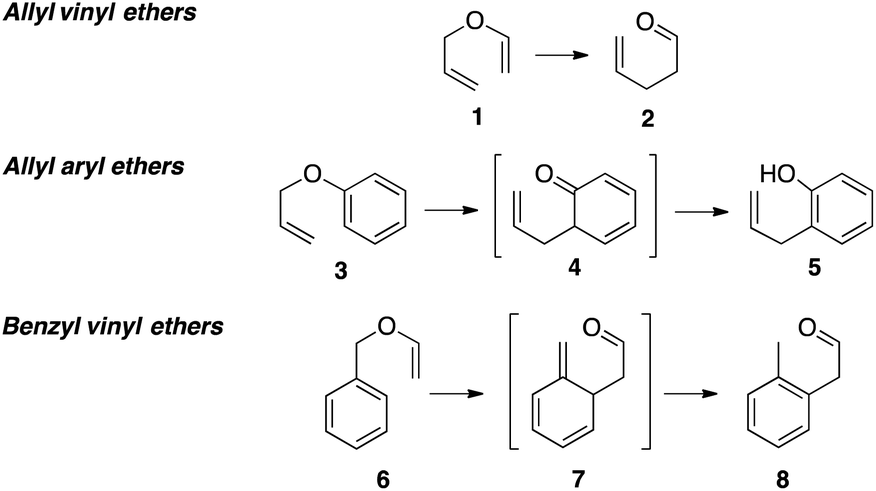
Claisen rearrangements of benzyl vinyl ethers: theoretical investigation of mechanism, substituent effects, and regioselectivity - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C7OB01666B

Allyl vinyl ethers via Wittig olefination: a short and efficient synthesis of (±)-mesembrine - ScienceDirect

Predict the major products of the reaction: Allyl vinyl ether is treated with HI in equimolar ratio - Chemistry - - 10642779 | Meritnation.com

Stereoselective Access to Highly Substituted Vinyl Ethers via trans-Difunctionalization of Alkynes with Alcohols and Iodine(III) Electrophile | Journal of the American Chemical Society

Claisen Rearrangement of Aliphatic Allyl Vinyl Ethers in the Presence of Copper(II) Bisoxazoline | The Journal of Organic Chemistry
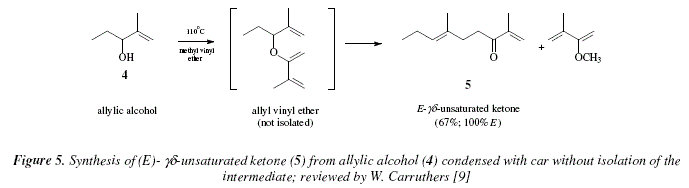
Synthesis of alkenes: claisen rearrangement of allyl vinyl ethers, part i; mechanistic views; the organic chemistry notebook series, a didactical approach, N2 9

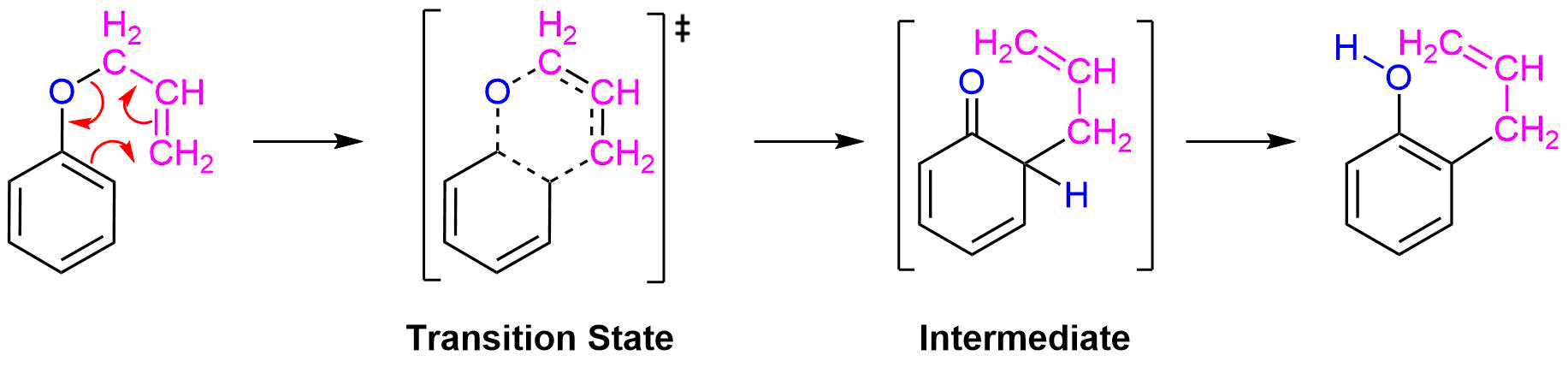
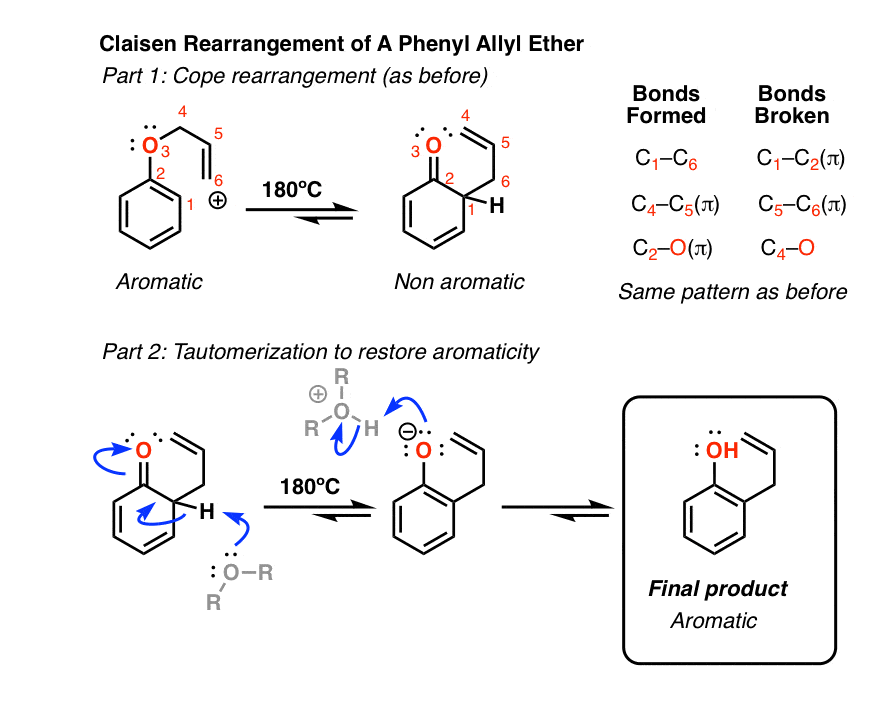
When a 2,6-disubstituted allyl phenyl ether is heated in an attempted Claisen rearrangement, migration occurs to give the p-allyl product as the result of two sequential pericyclic reactions. Explain. | Homework.Study.com
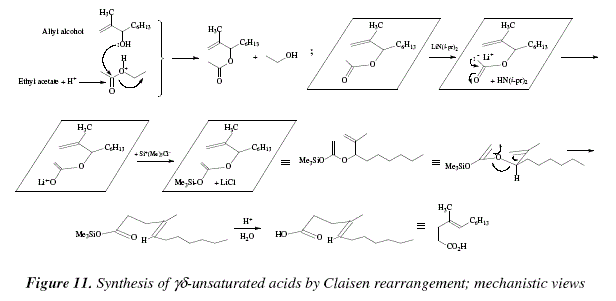
Synthesis of alkenes: claisen rearrangement of allyl vinyl ethers, part i; mechanistic views; the organic chemistry notebook series, a didactical approach, N2 9
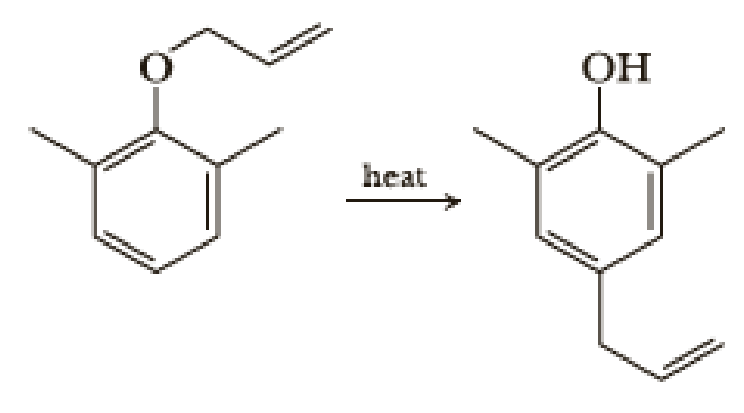
Claisen rearrangement of an allyl phenyl ether with substituent groups in both ortho positions leads to the formation of a para-substituted product. Propose a mechanism for the following rearrangement. | bartleby