Commission notice – Application of the Union's pharmaceutical acquis in markets historically dependent on medicines supply f
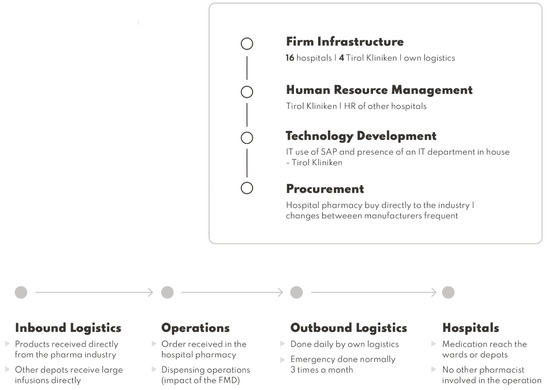
IJERPH | Free Full-Text | Falsified Medicines Directive in a Secondary Care Environment—Impact on Supply Chain

Regional empowerment through decentralised governance under a centralised regulatory system facilitates the development of cellular therapy in China - The Lancet Haematology
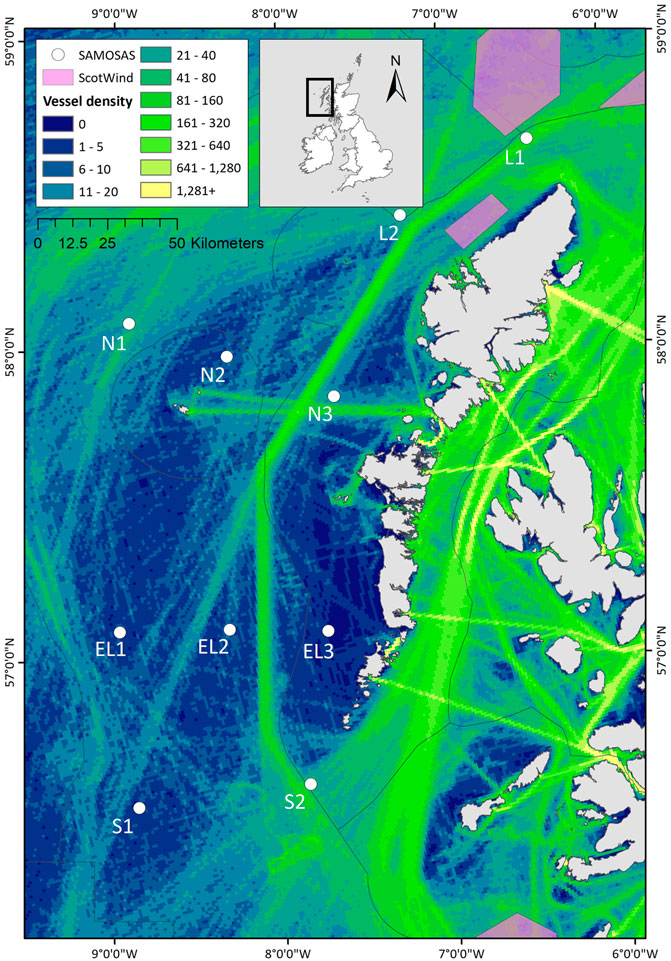
Frontiers | Monitoring cetacean occurrence and variability in ambient sound in Scottish offshore waters
ESSD - The consolidated European synthesis of CH4 and N2O emissions for the European Union and United Kingdom: 1990–2019
Commission Delegated Regulation (EU) 2016/161 of 2 October 2015 supplementing Directive 2001/83/EC of the European Parliament an
EUROPEAN COMMISSION DIRECTORATE-GENERAL FOR HEALTH AND FOOD SAFETY Health systems and products Medical products – quality, saf
Commission ImplementingRegulation (EU) 2019/ of 9 October 2019 amending Annex I to Council Regulation (EEC)No 2658/87 on the ta
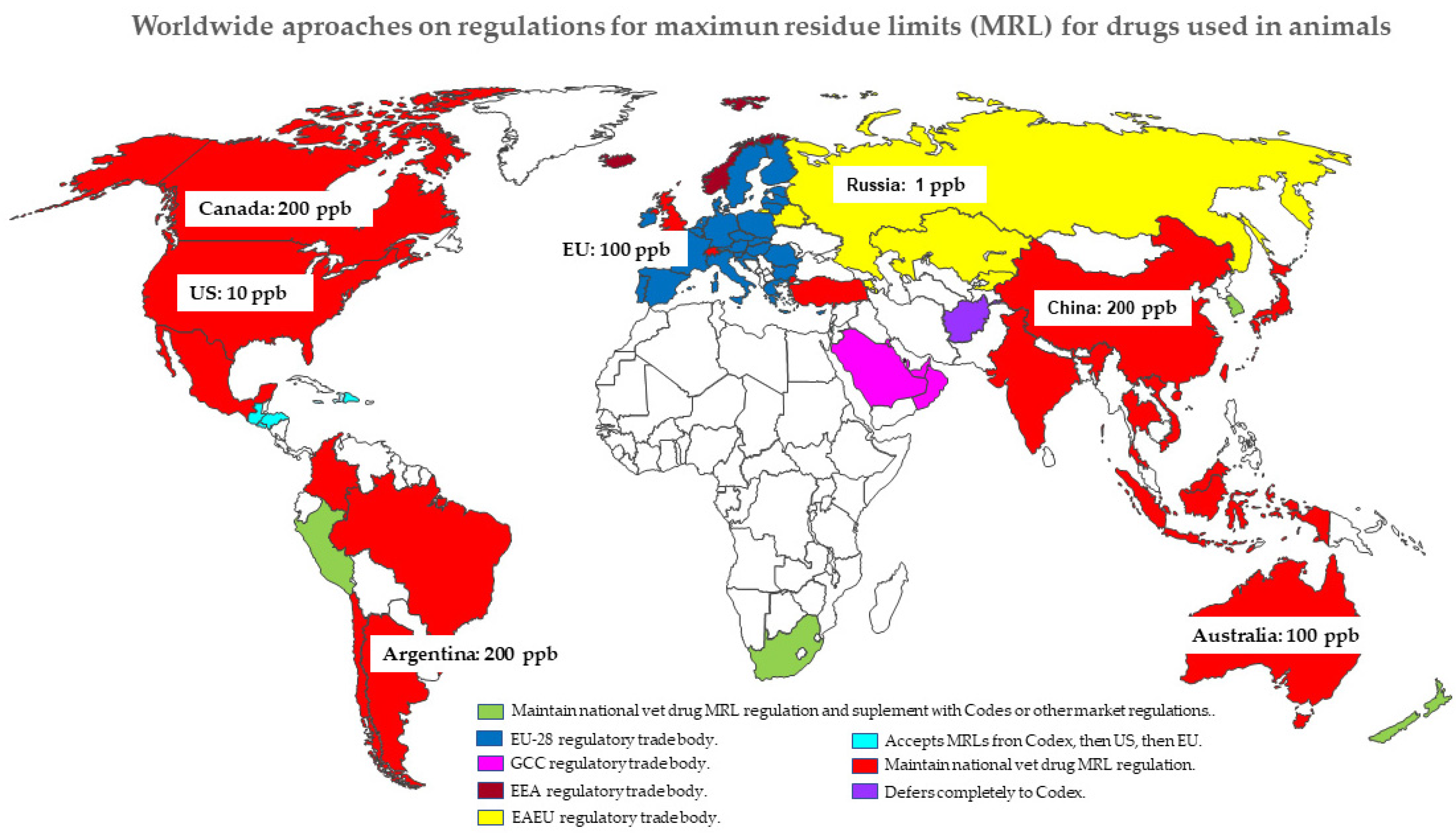
Animals | Free Full-Text | Rational Pharmacotherapy in Infectious Diseases: Issues Related to Drug Residues in Edible Animal Tissues
AIDE MEMOIRE FOR GDP INSPECTION OF WHOLESALERS COMPLIANCE WITH COMMISSION DELEGATED REGULATION (EU) 2016/161 FOR SAFETY FEATURE









