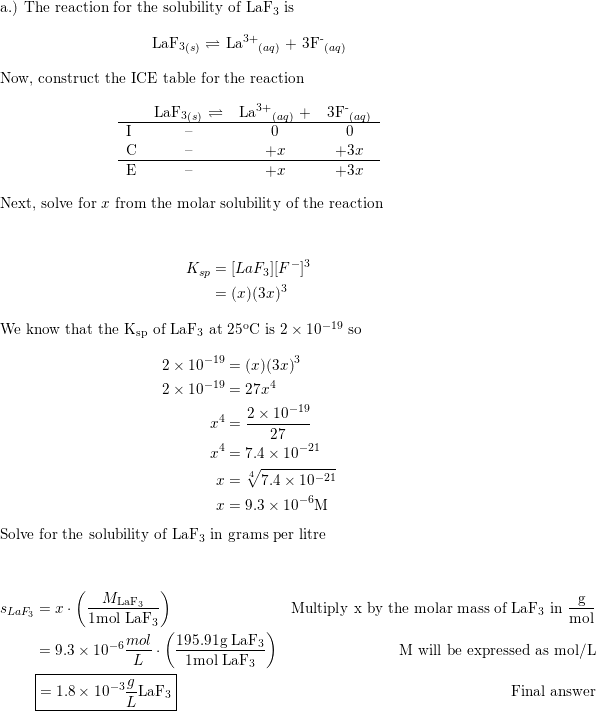Concentration. Concentration Particles per volume Can be in grams per litre but chemists usually express concentration in moles per litre This are related. - ppt download

The amount of salt, in grams, in a solution is proportional to the volume, in liters, of the solution as - Brainly.com

The solubility product of chalk is `9.3xx10^(-8)`. Calculate its solubility in gram per litre - YouTube

SOLVED: Calculate the density of SOz gas, in grams per liter; at 55*C and 1.5 atm (MWs-32 g/mole, MWo-16 g/mole) 3.2 g/L 3.4g/L 3.6 g/L 3.89/L None of the above

SOLVED:Calculate the solubility of each of the following compounds in moles per liter and grams per liter. (Ignore any acid-base properties.) a. A g3 P O4, Ks p=1.8 ×10^-18 b. CaCO3, Ksp=8.7 ×

The amount of salt, in grams, in a solution is proportional to the volume, in liters, of the solution as - Brainly.com

The density of a certain material is such that it weighs 6 pounds per pint of volume. Express this density - Brainly.com




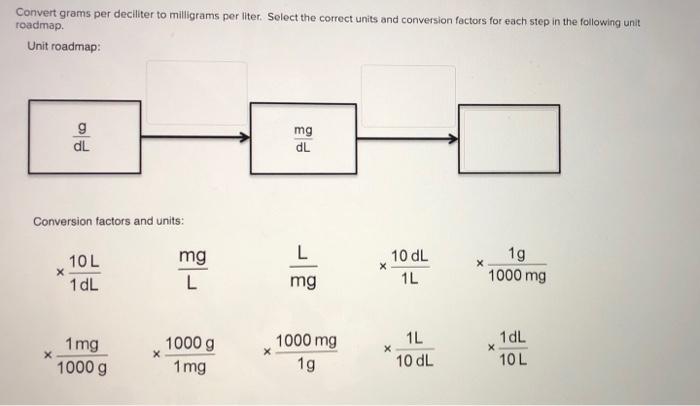

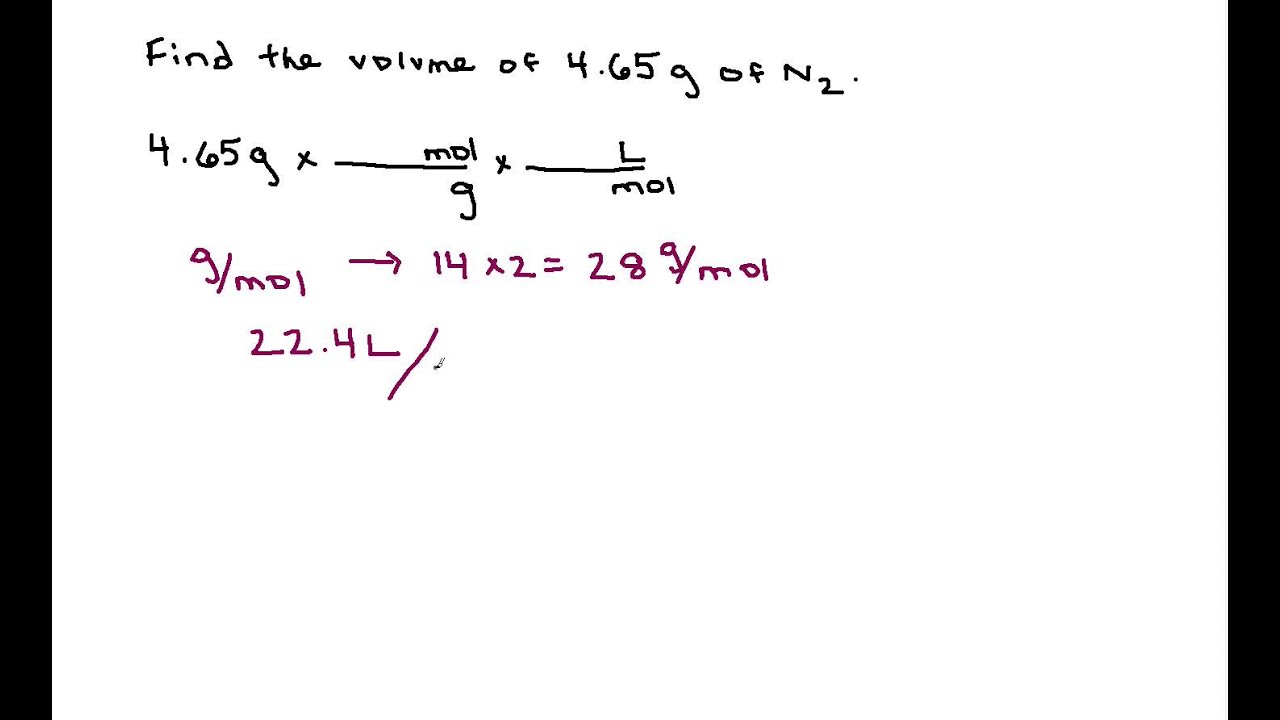







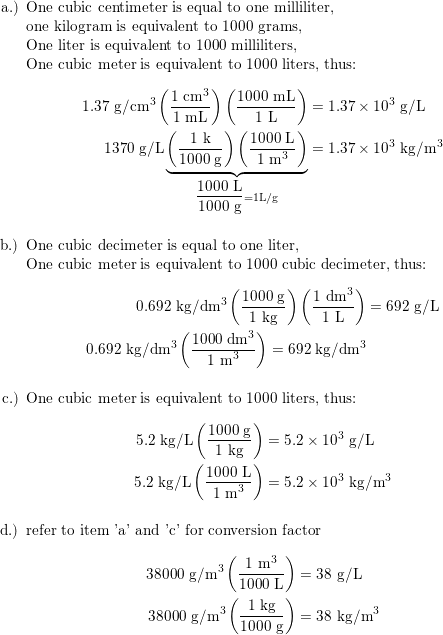


:max_bytes(150000):strip_icc()/how-to-calculate-normality-609580final2-0d5efa5a961f4fa0a7efc780921faee1.png)