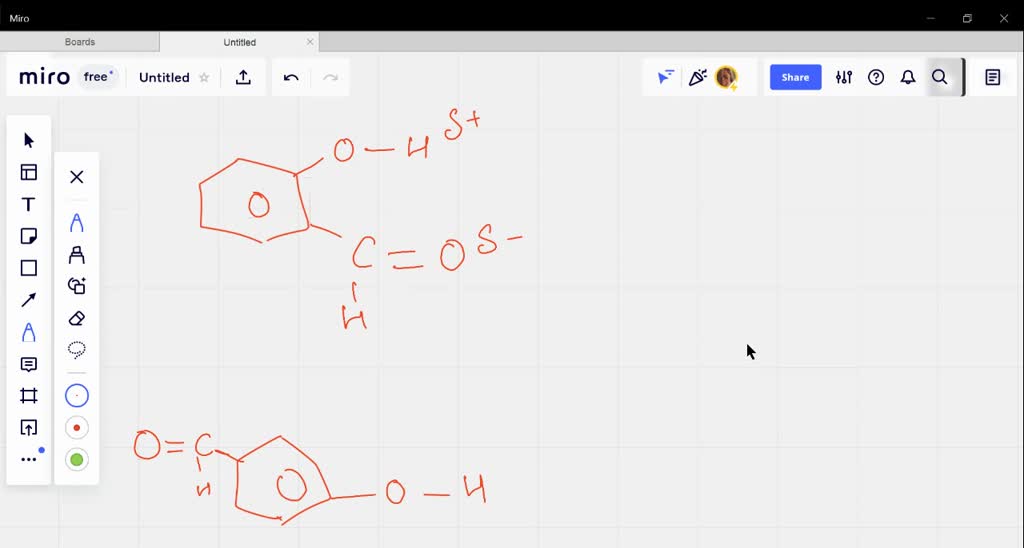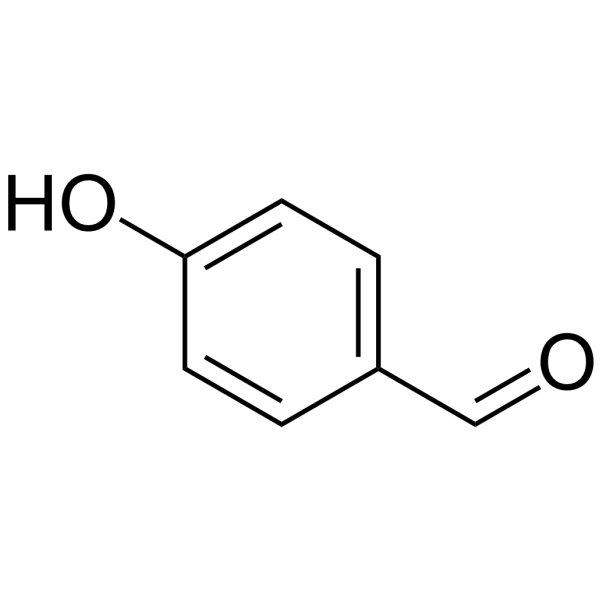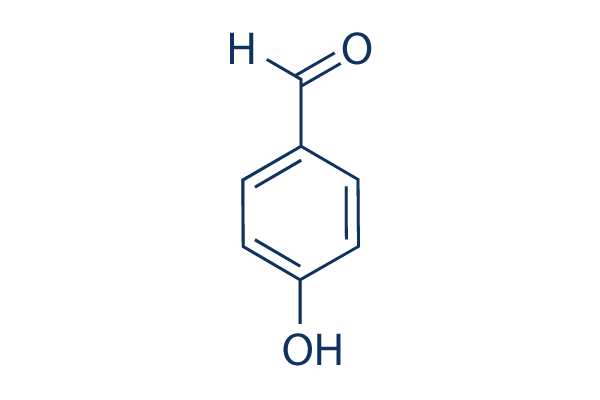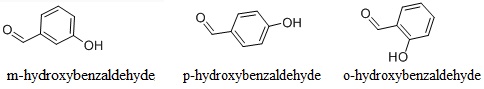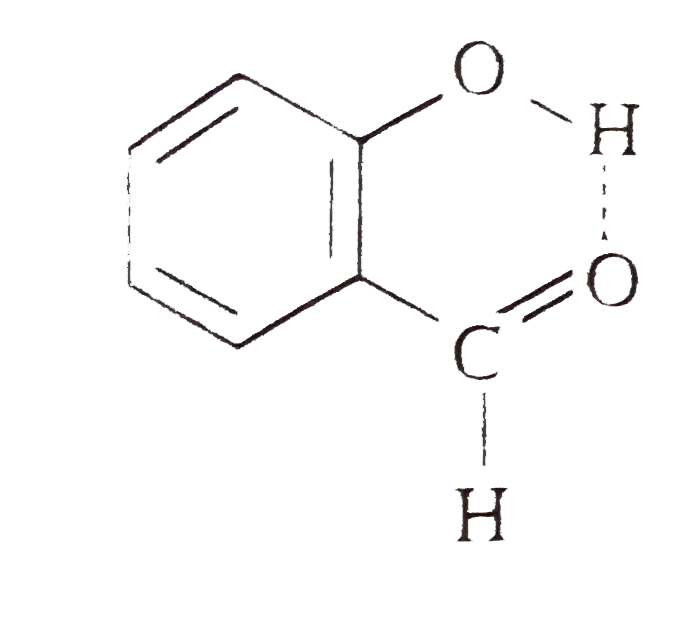
Explain why is o-hydroxybenzaldehyde a liquis at room temperaturre while p- hydroxybenzaldehyde is a high melting solid.

o-Hydroxy benzaldehyde is more soluble in water than p-hybroxy benzaldehde (b) o-Hydroxy b - YouTube
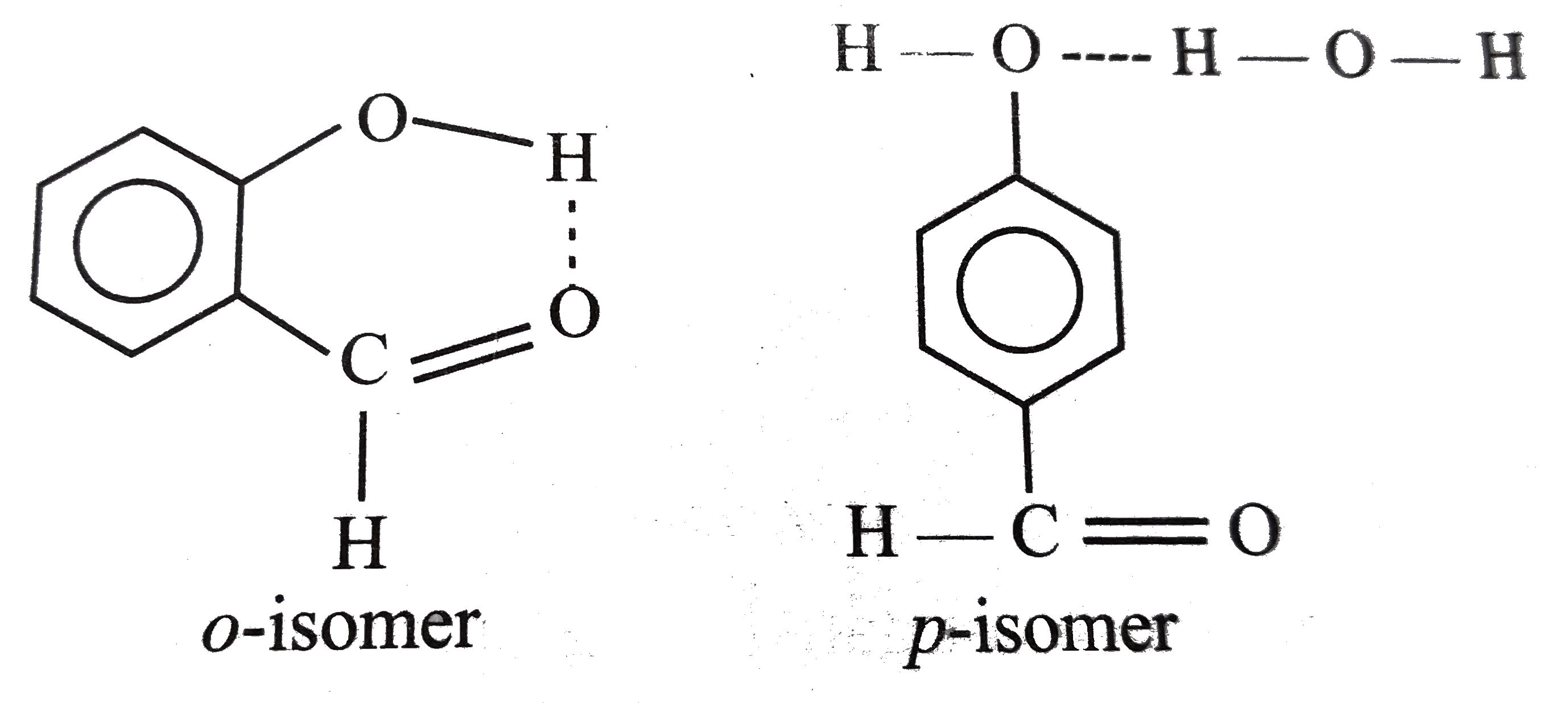
o-Hydroxy benzaldehyde is more soluble in water than p-hybroxy benzaldehde (b) o-Hydroxy benzaldehyde is liquid at room temperature while p-hydroxy benzaldehyde is high melting solid .
Explain; why o-hydroxy benzaldehyde is a liquid at room temperature while p-hydroxy benzaldehyde is a high melting solid ? - Sarthaks eConnect | Largest Online Education Community

When benzene in presence of anhydrous aluminium chloride reacts with ethyl chloride the compound formed is:
![PDF] Tuning the Strength of the Resonance-Assisted Hydrogen Bond in o- Hydroxybenzaldehyde by Substitution in the Aromatic Ring1. | Semantic Scholar PDF] Tuning the Strength of the Resonance-Assisted Hydrogen Bond in o- Hydroxybenzaldehyde by Substitution in the Aromatic Ring1. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b3991e25b71615eec049e2836fc71828f431c91a/3-Figure2-1.png)
PDF] Tuning the Strength of the Resonance-Assisted Hydrogen Bond in o- Hydroxybenzaldehyde by Substitution in the Aromatic Ring1. | Semantic Scholar

1649426-95-8 | Salicylaldehyde-13C6 | Salicylal-13C6; 2-Formylphenol-13C6; 2-Hdroxybenzaldehyde-13C6; 2-Hydroxybenzaldehyde-13C6; NSC 112278-13C6; NSC 49178-13C6; NSC 83559-13C6; NSC 83560-13C6; NSC 83561-13C6; NSC 83562-13C6; NSC 97202-13C6; Salicylal ...

Explain, why o-hydroxybenzaldehyde is a liquid at room temperature while p- hydroxybenzaldehyde is a high melting solid?

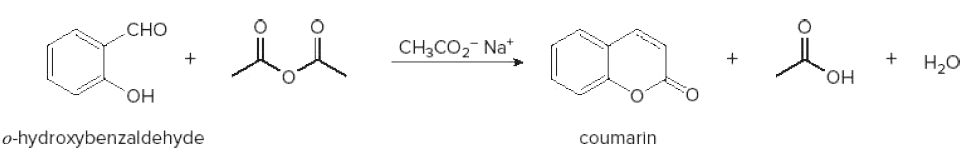
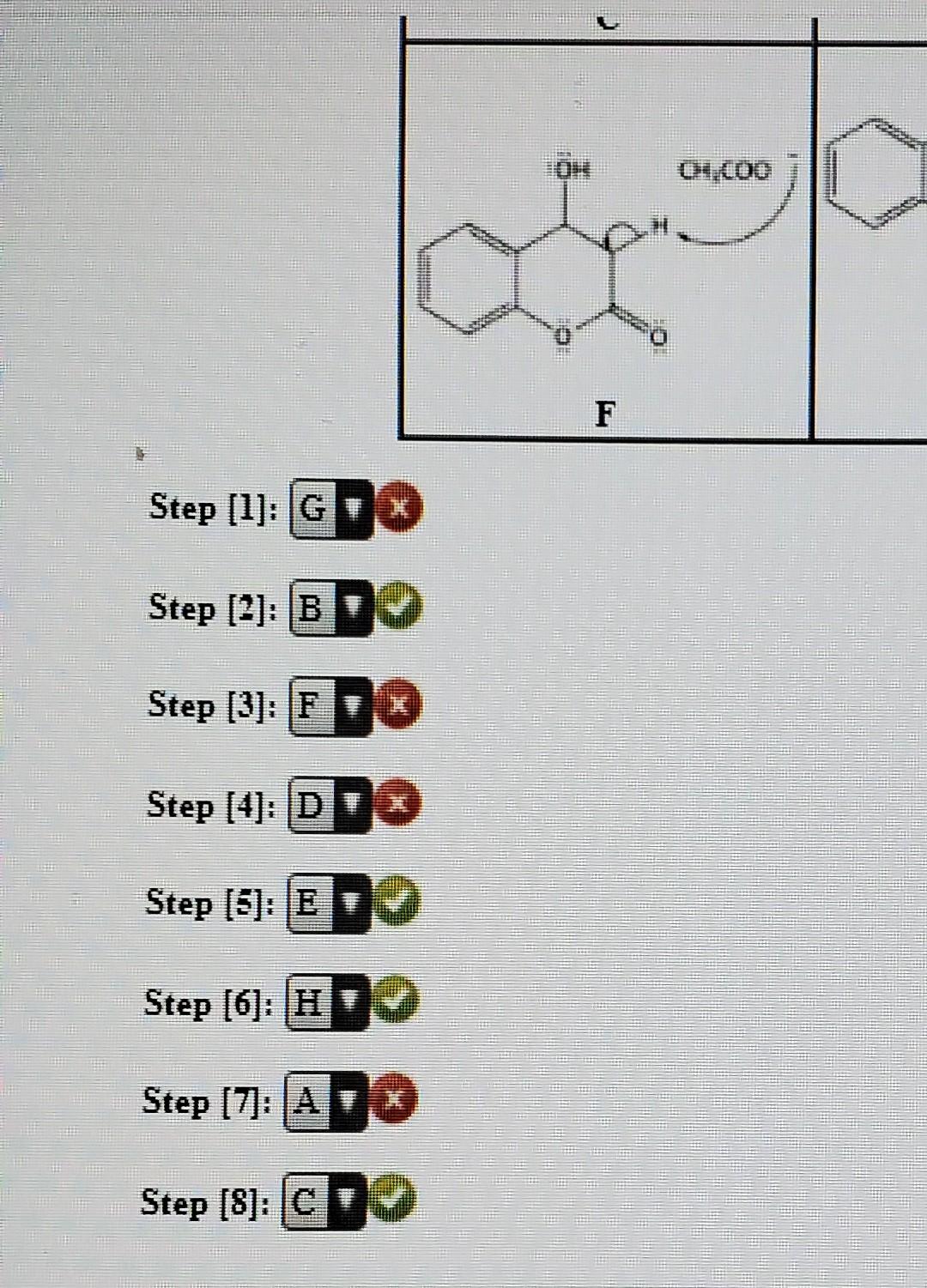
When 0 hydroxybenzaldehyde is heated with ethanoic an hydride in presence sodium ethanoate, compound formed during the reaction is ?

Structural and orthoselectivity study of 2-hydroxybenzaldehyde using spectroscopic analysis - ScienceDirect
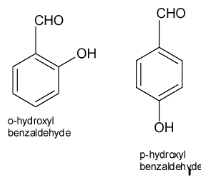
Assertion: Both o-hydroxybenzaldehyde and p-hydroxy benzaldehyde have the same molecular weight and show H-bonding.Reason: Melting point of p- hydroxybenzaldehyde is more than o-hydroxybenzaldehyde.A. Both assertion and reason are correct and reason is the

o - Hydroxybenzaldehyde is a liquid at room temperature while p - hydroxybenzaldehyde is a high melting solid because of: