Identifying the Rate Law for a Reaction from a Mechanism Where the First Step is Rate Limiting | Chemistry | Study.com

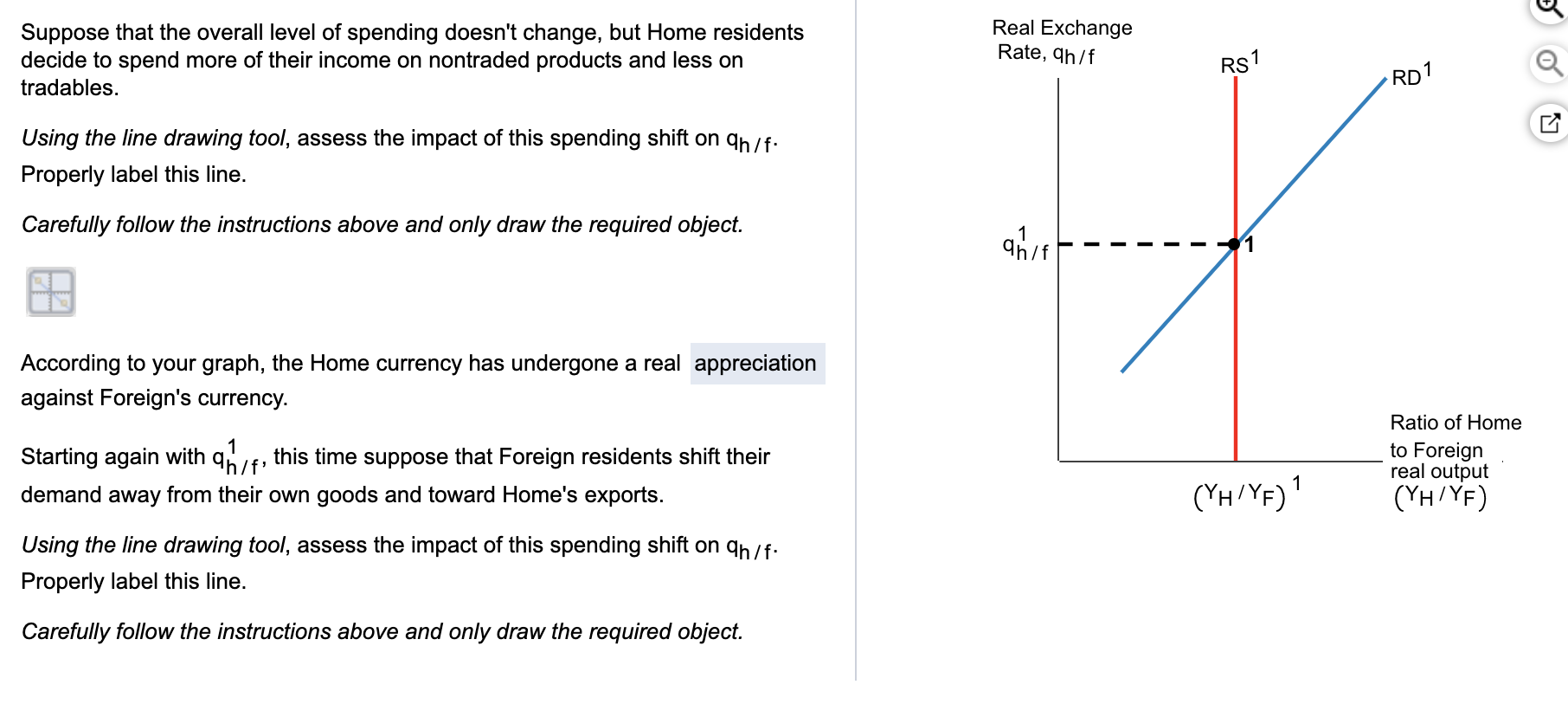
1) A decrease in the overall level of investment in an economy, holding everything else constant, causes the price level (as evidenced by the CPI or GDP Deflator) to......in the short run

SOLVED: Consider the mechanism Step I: A = B+C slow Step 2: C+D -E Overall: A+D - B+E Determine the rate law for the overall reaction, where the overall rate constant is
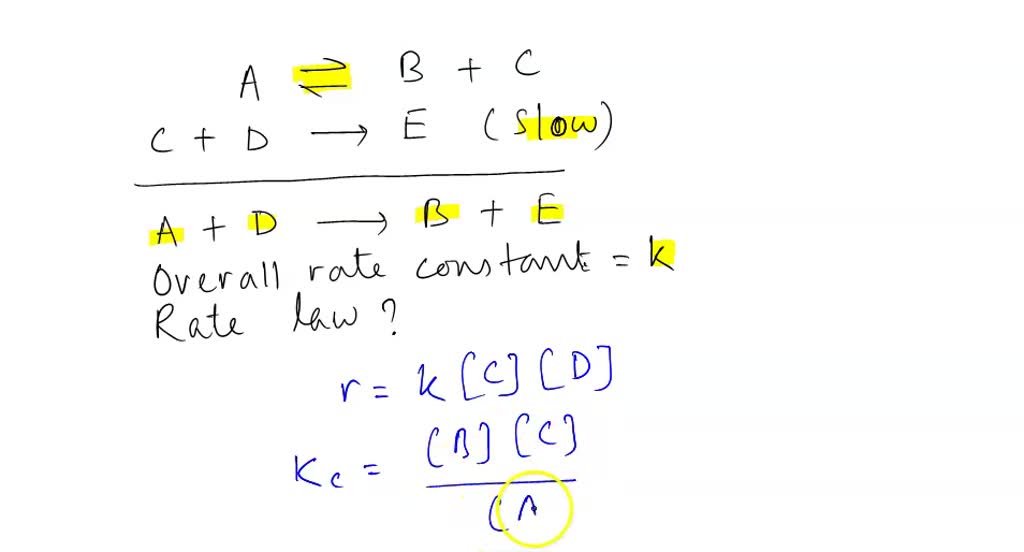
SOLVED: Consider the mechanism. Step 1:A↽⇀B+C equilibrium Step 2: C+D⟶E slow :Overall: A+D⟶B+E Determine the rate law for the overall reaction, where the overall rate constant is represented as k.
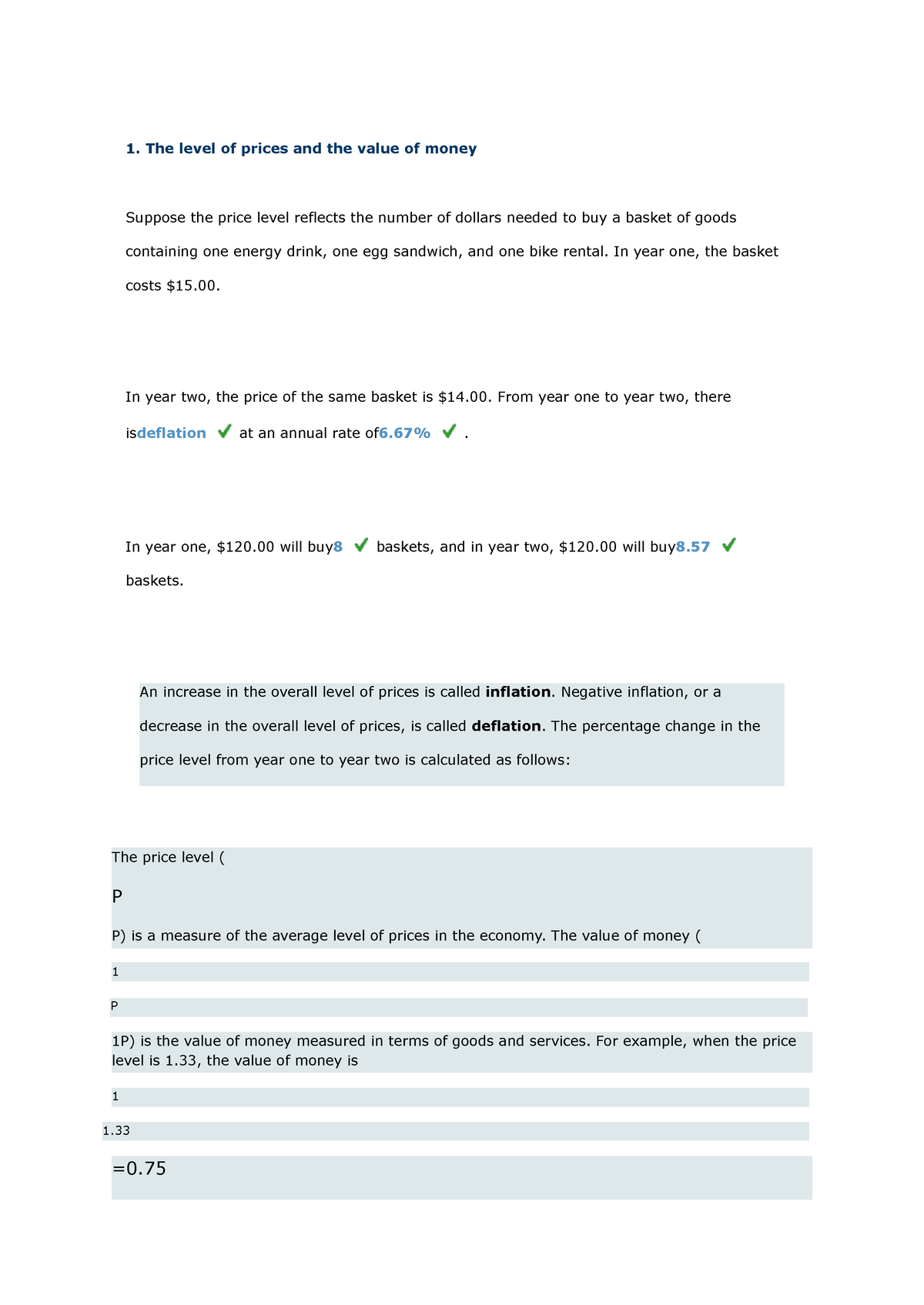
1 - history - 1. The level of prices and the value of money Suppose the price level reflects the - Studocu

what effect does a decrease in temperature have on the overall rate of a chemical reaction?? A decreased in - Brainly.com

For a complex reaction A k products Ea1 = 180 kJ/mol ; Ea2 = 80 kJ/mol ; Ea3 = 50 kJ/mol Overall rate constant k is related to individual rate constant by

Determine the rate law for the overall reaction (where the overall rate constant is represented as k)? | Socratic

:max_bytes(150000):strip_icc()/Modern-Portfolio-V2-9a0e2a7c92764f0cb194615eaedcdd76.jpg)