PDF) Association Between Participation in Clinical Trials and Overall Survival Among Children With Intermediate- or High-risk Neuroblastoma

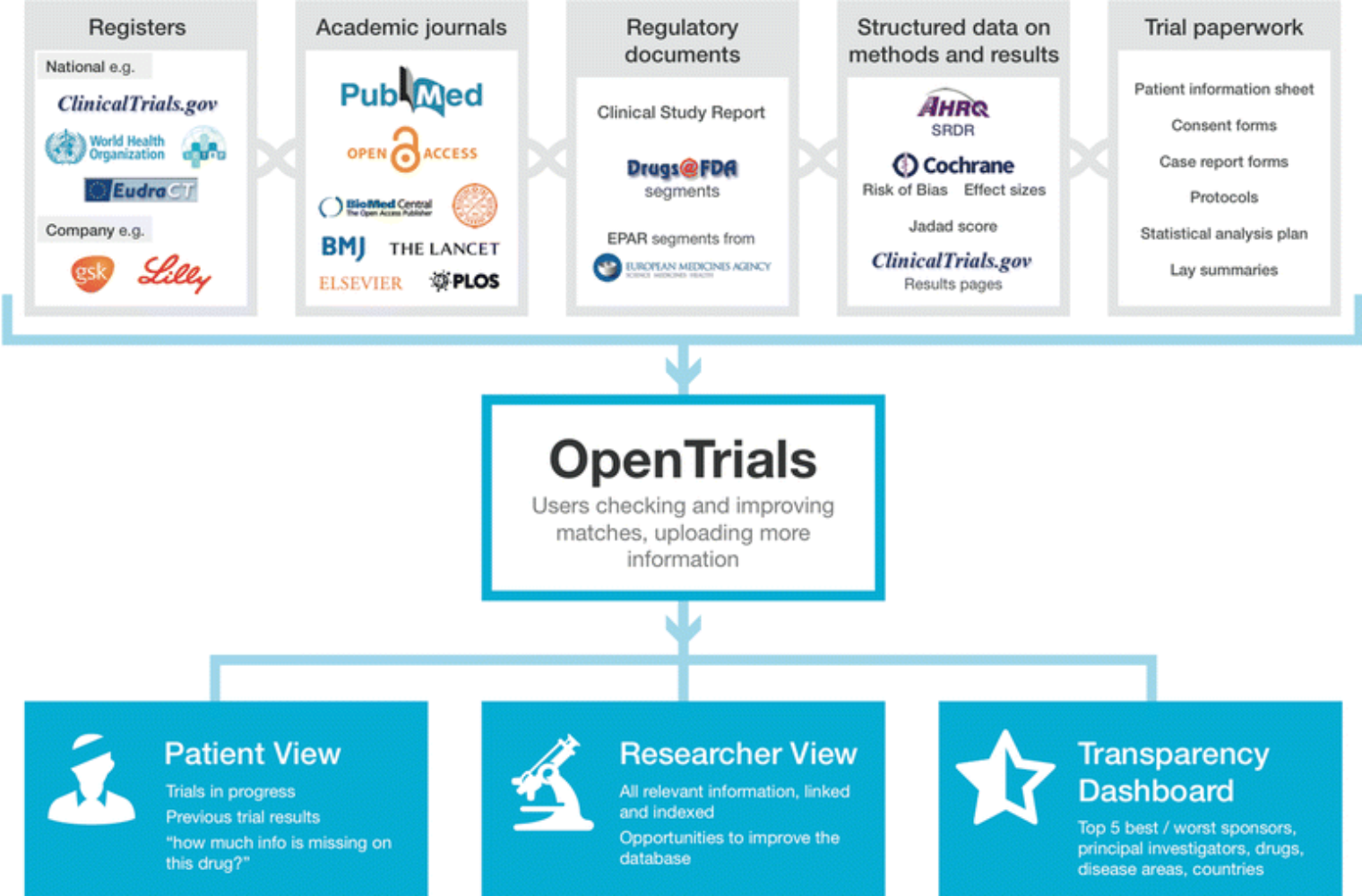
Advanced Analytics is revolutionizing clinical trials: the experience of Exom Group, the first European digital CRO, to implement such an innovative approach. | Exom Group

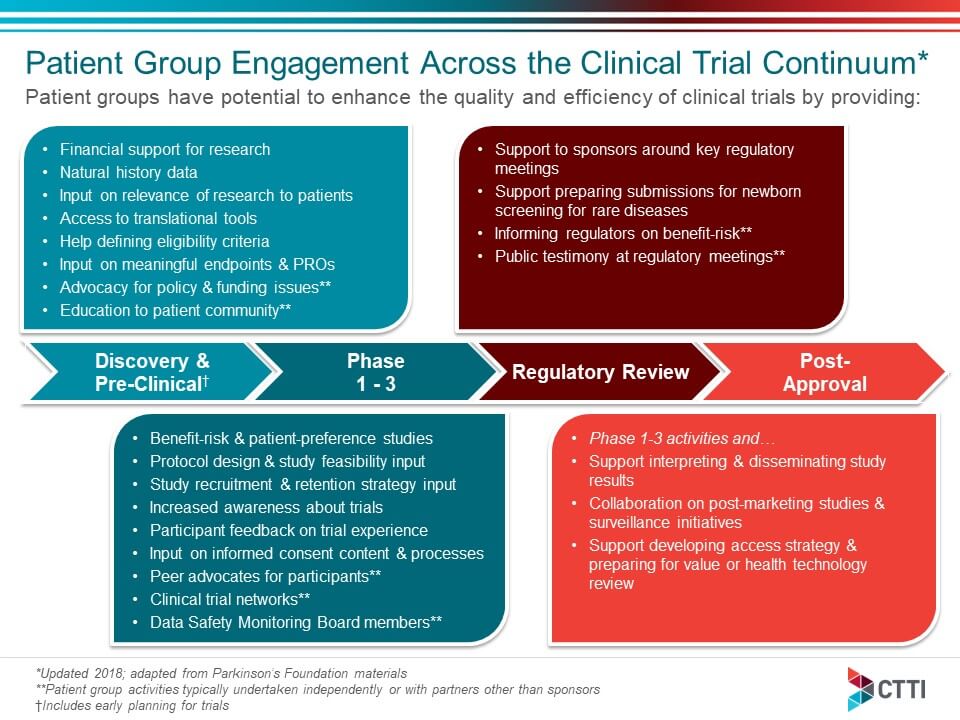
A Glimpse Into The Clinical Trial Participant ExperienceAs Designed By The Pharmaceutical Industry - DT Consulting

A Glimpse Into The Clinical Trial Participant ExperienceAs Designed By The Pharmaceutical Industry - DT Consulting

A Glimpse Into The Clinical Trial Participant ExperienceAs Designed By The Pharmaceutical Industry - DT Consulting

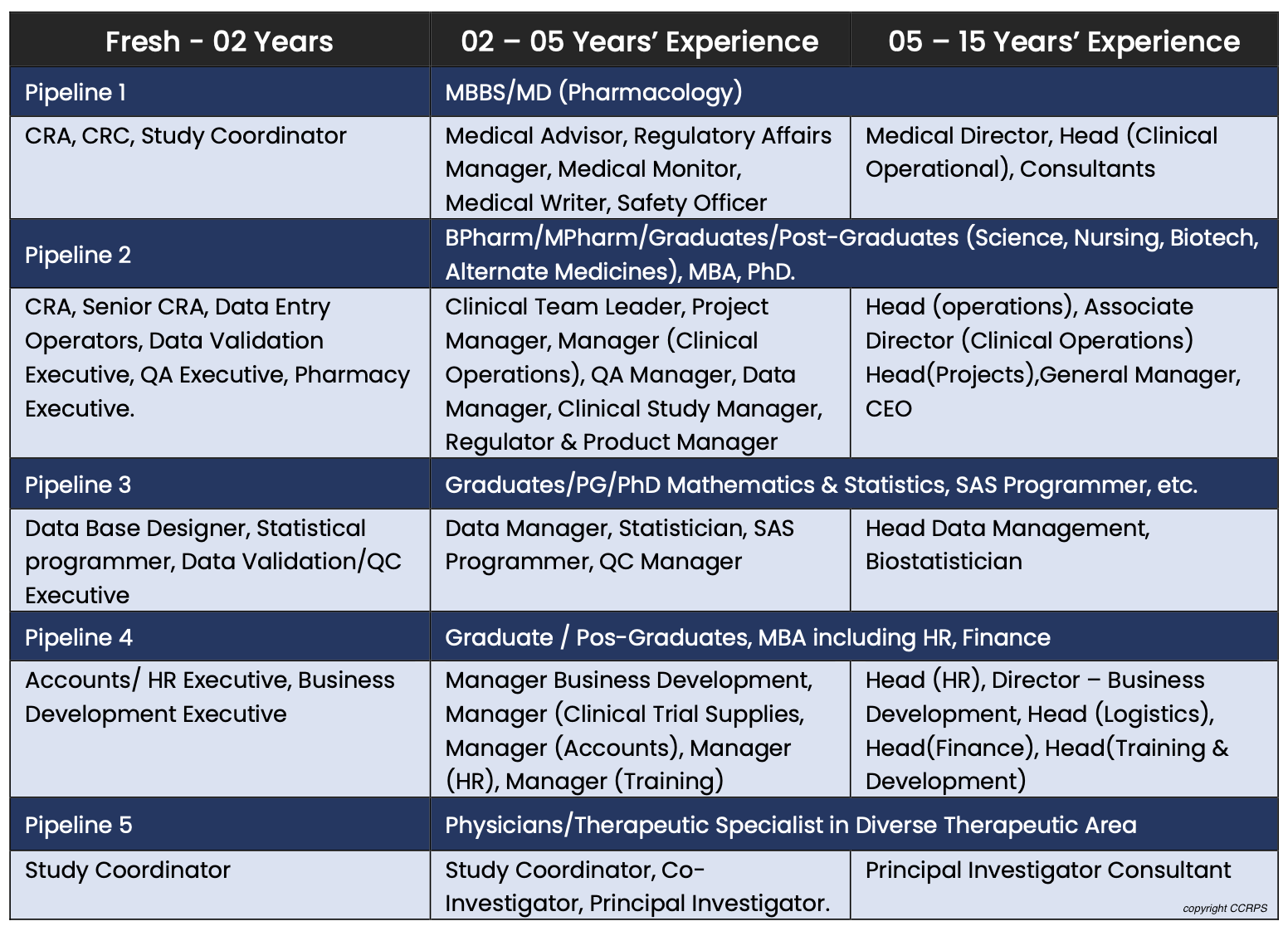
The role of the clinical research coordinator – data manager – in oncology clinical trials – topic of research paper in Clinical medicine. Download scholarly article PDF and read for free on

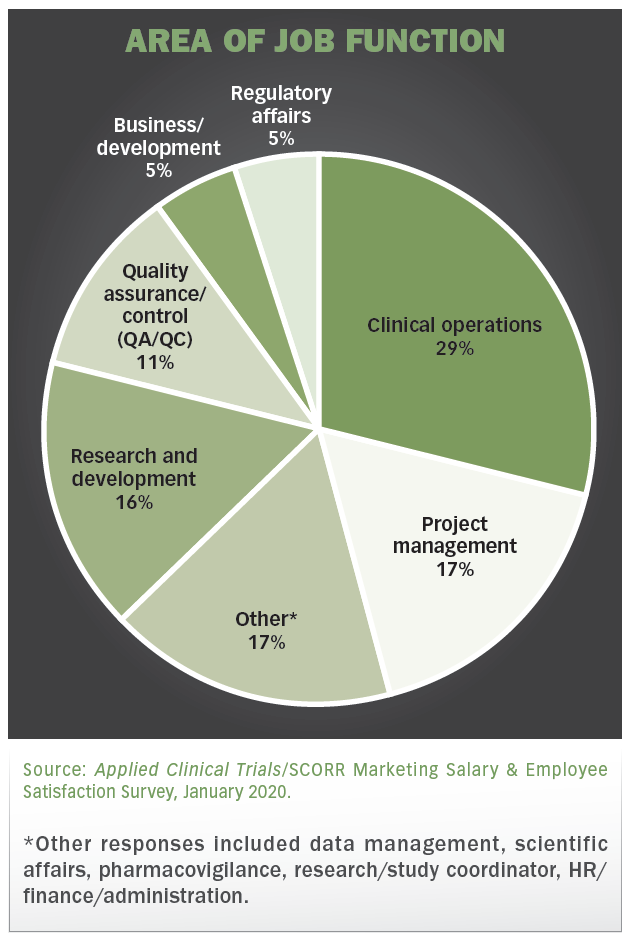
PDF) How a Clinical Trial Unit can improve independent clinical research in rare tumors: the Italian Sarcoma Group experience