
Cancers | Free Full-Text | Anti-Tumor Effect of Inhibition of DNA Damage Response Proteins, ATM and ATR, in Endometrial Cancer Cells
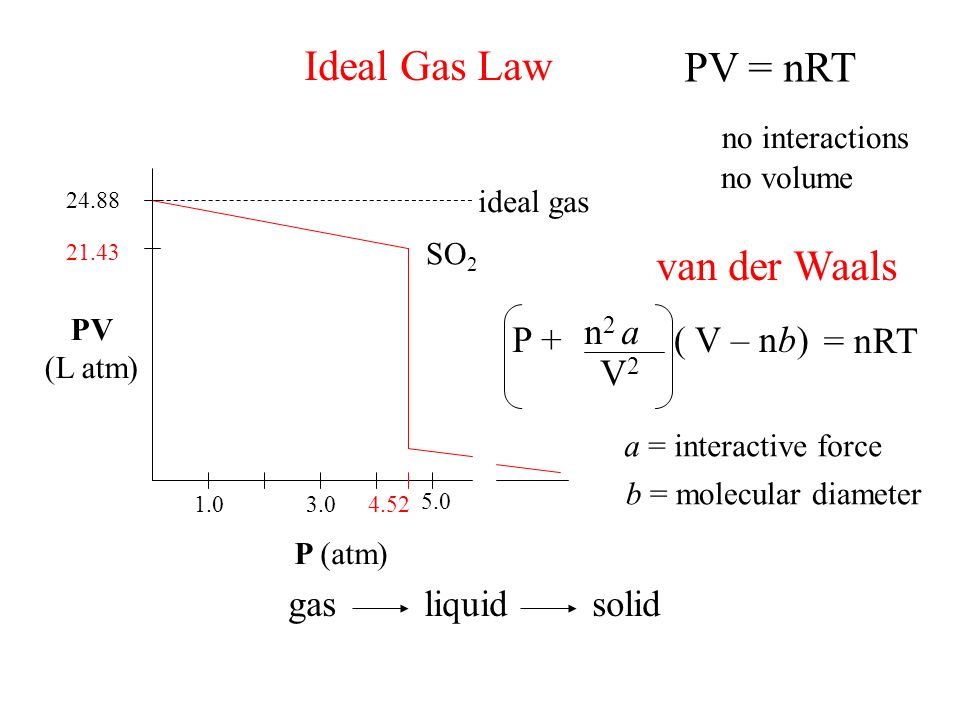
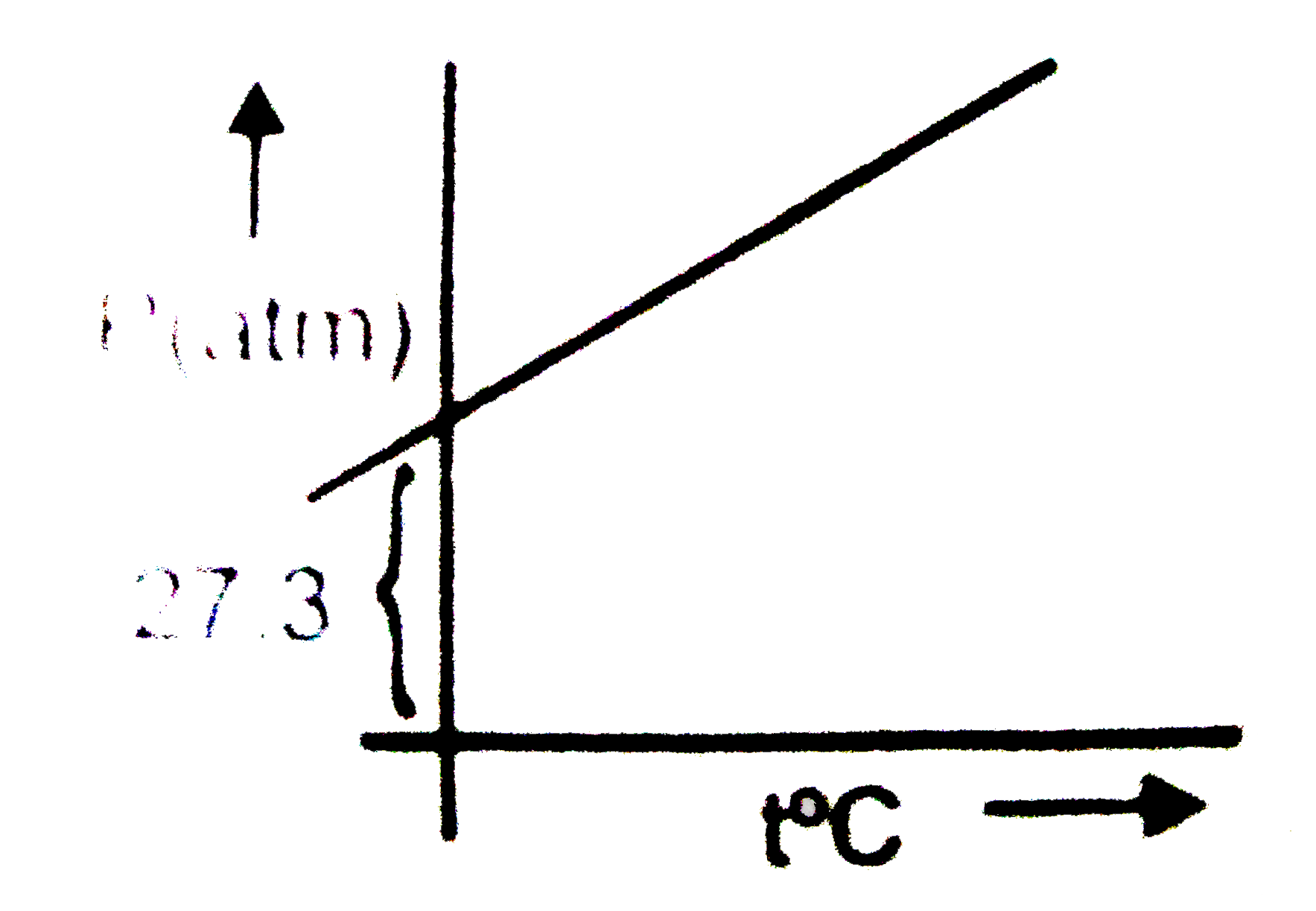
Ideal Gas Law PV = nRT PV (L atm) P (atm) ideal gas SO no volume no interactions van der Waals P + n 2 a V2V2 ( V – nb) = - ppt download

A) Western blot analysis for expression of phosphorylated ATM (pATM),... | Download Scientific Diagram
![The pressure P [atm] as a function of temperature T [K] for different... | Download Scientific Diagram The pressure P [atm] as a function of temperature T [K] for different... | Download Scientific Diagram](https://www.researchgate.net/publication/304531206/figure/fig7/AS:668377173475329@1536364944027/The-pressure-P-atm-as-a-function-of-temperature-T-K-for-different-number-densities-n.png)
The pressure P [atm] as a function of temperature T [K] for different... | Download Scientific Diagram

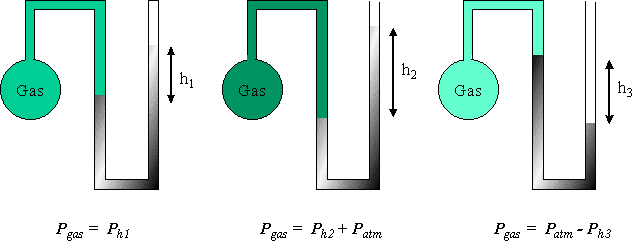
Chapter 12 Gases. What is the pressure of the gas in the bulb? 1. P gas = P h 2. P gas = P atm 3. P gas = P h +
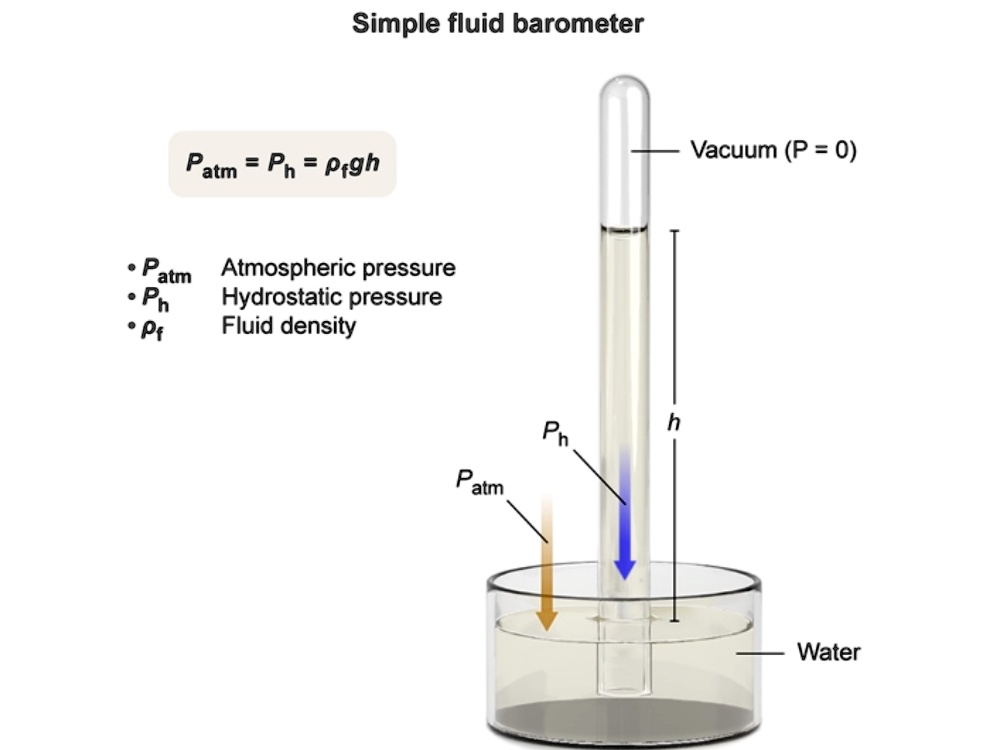
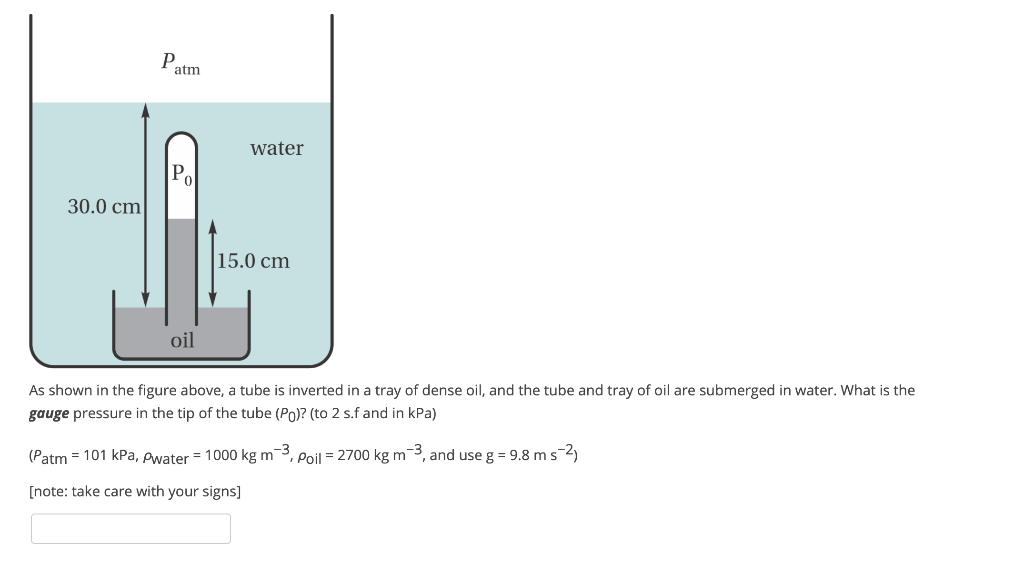
SOLVED: Simple fluid barometer Vacuum (P = 0) Patm Ph Prgh Patm Atmospheric pressure Ph Hydrostatic pressure Pf Fluid density Ph P atm Water

Rishi Kumar, MD - Atmospheric pressure (P,atm) is ~760 mmHg at sea level. Alveolar pressure (P,alv) is the pressure within the alveolus. The intrapleural space is between the visceral pleura (a serous

JCI - ATM inhibition enhances cancer immunotherapy by promoting mtDNA leakage and cGAS/STING activation
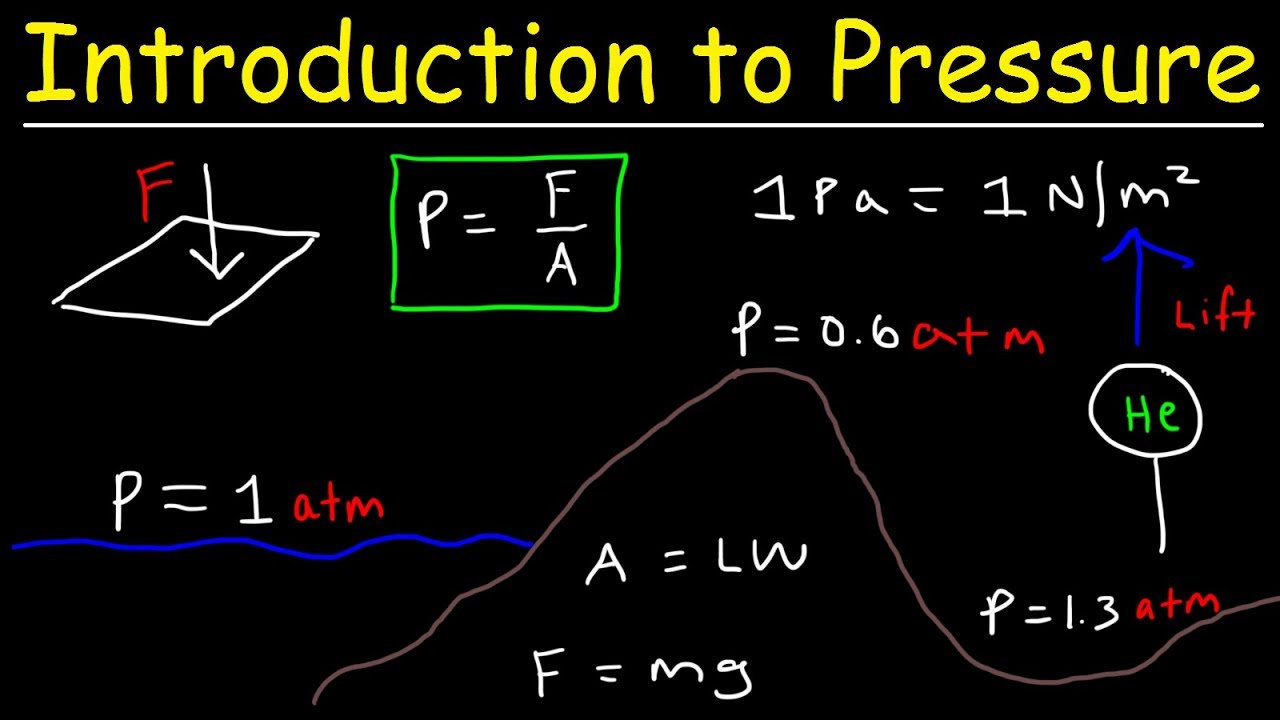
Introduction to Pressure - Force & Area, Units, Atmospheric Gases, Elevation & Boiling Point - YouTube

SOLVED:What is the mass density of air at P=1.0 atm and T=(a)-10^∘ C and (b) 30^∘ C ? The average molecular mass of air is approximately 29 u.
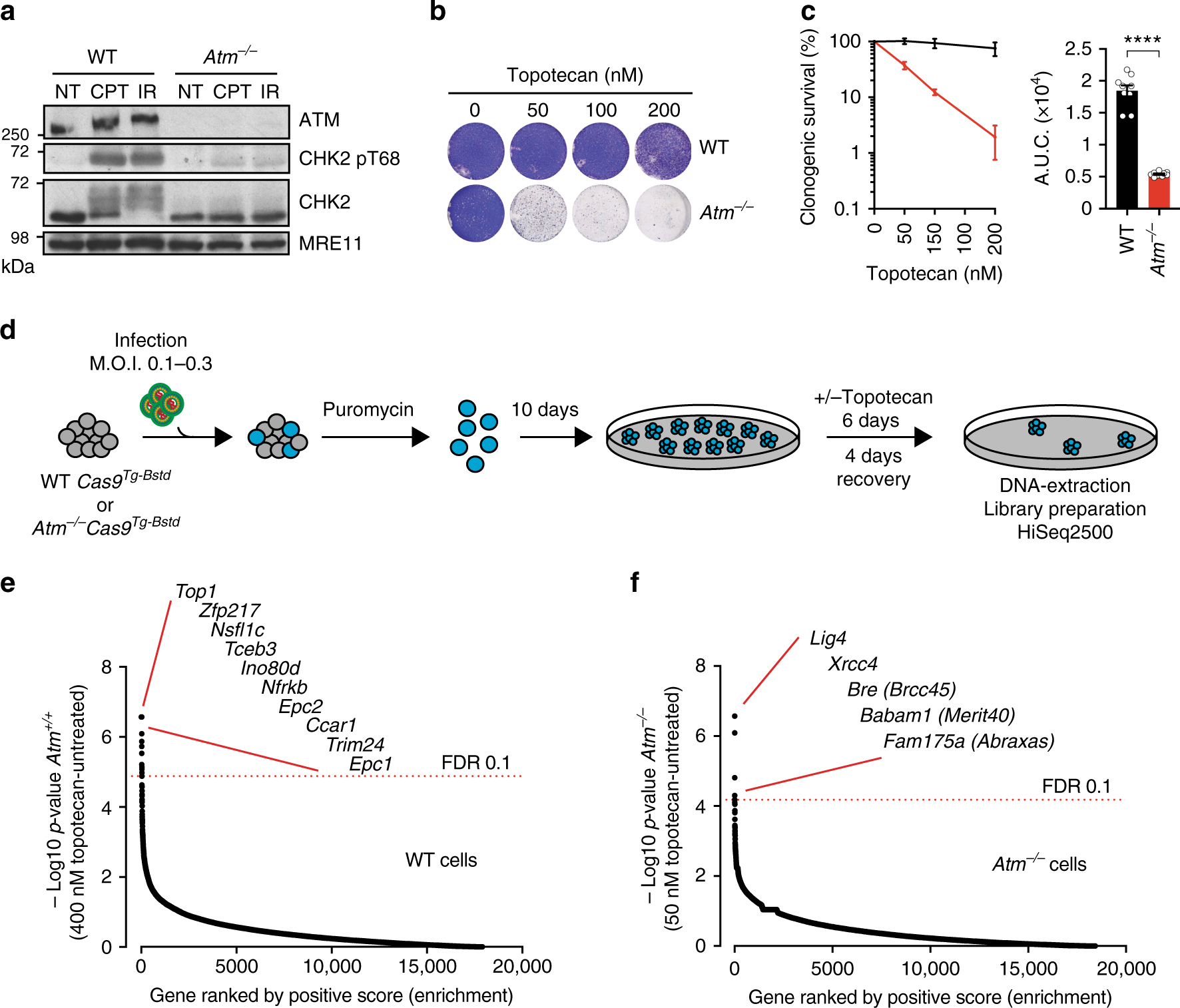
ATM orchestrates the DNA-damage response to counter toxic non-homologous end-joining at broken replication forks | Nature Communications
![SOLVED: [References] Convert the pressure to atm: 1 atm P = 370. mm Hg X 760 mm Hg )= 0.487 atm Determine the amount of ClF3= (0.487 atm)(1.5 L) 0.030 (0.082057 L SOLVED: [References] Convert the pressure to atm: 1 atm P = 370. mm Hg X 760 mm Hg )= 0.487 atm Determine the amount of ClF3= (0.487 atm)(1.5 L) 0.030 (0.082057 L](https://cdn.numerade.com/ask_images/b34acda3d42047b09ba5560f755ebabb.jpg)




![ATM [p Ser1981] Antibody (10H11.E12) (NB100-306): Novus Biologicals ATM [p Ser1981] Antibody (10H11.E12) (NB100-306): Novus Biologicals](https://images.novusbio.com/images2/ATM-[p-Ser1981]-Antibody-(10H11.E12)-Western-Blot-NB100-306-img0004.jpg)

