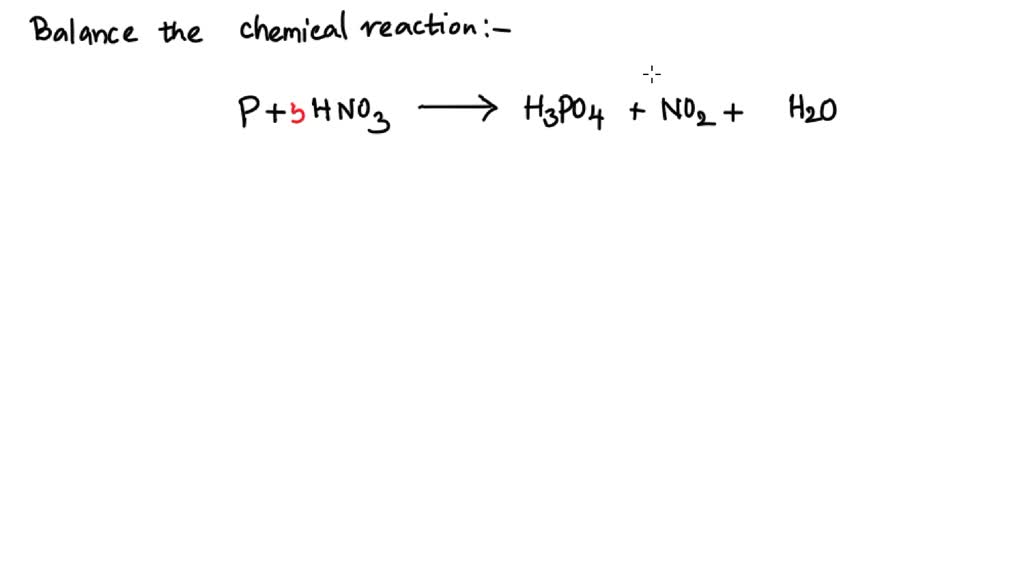
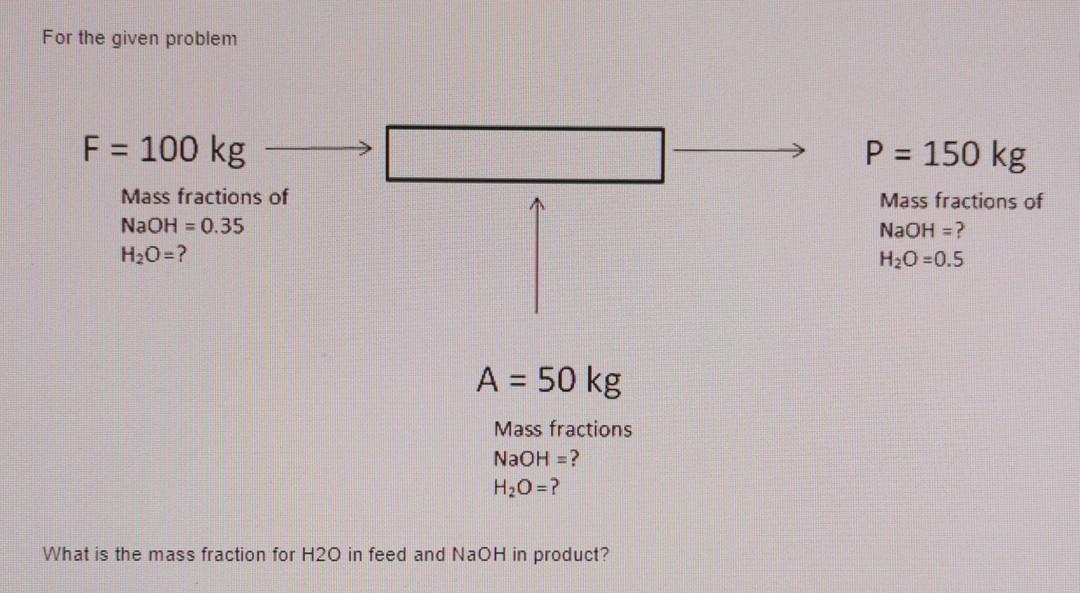
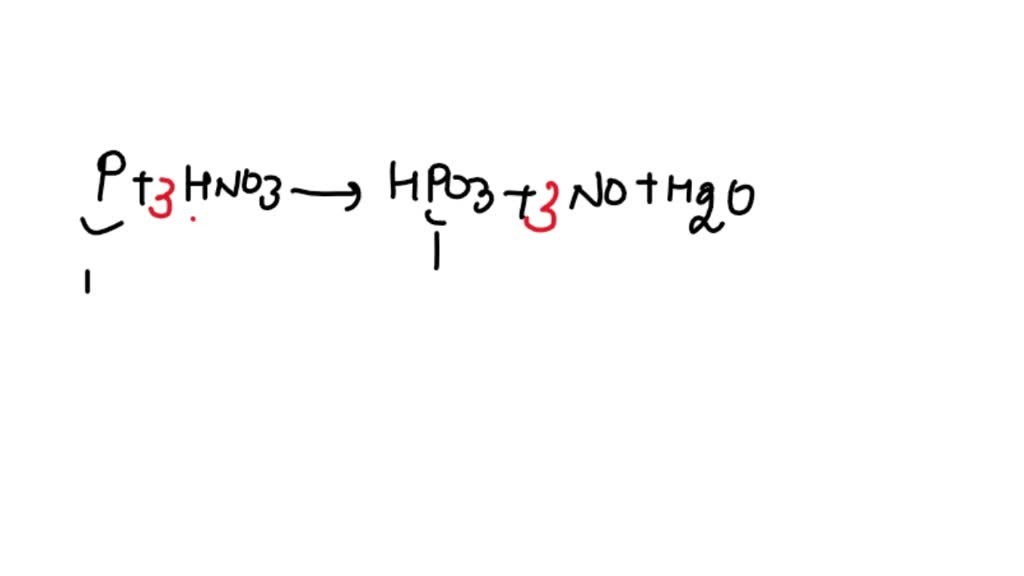ntWhich of the following compound gives p cresol with p methyl diazonium chloride?n nt1) H2O 2) H3PO2 3) HCOOH 4) C6H5OHn

P( 2 D) LIF signal following PLP of PCl3 at a total pressure of 7.64... | Download Scientific Diagram

File:3. p-nitrofenil-alfa-D-manopiranosídeo + H2O → p-nitrofenol + alfa-D-manopiranose.png - Wikimedia Commons
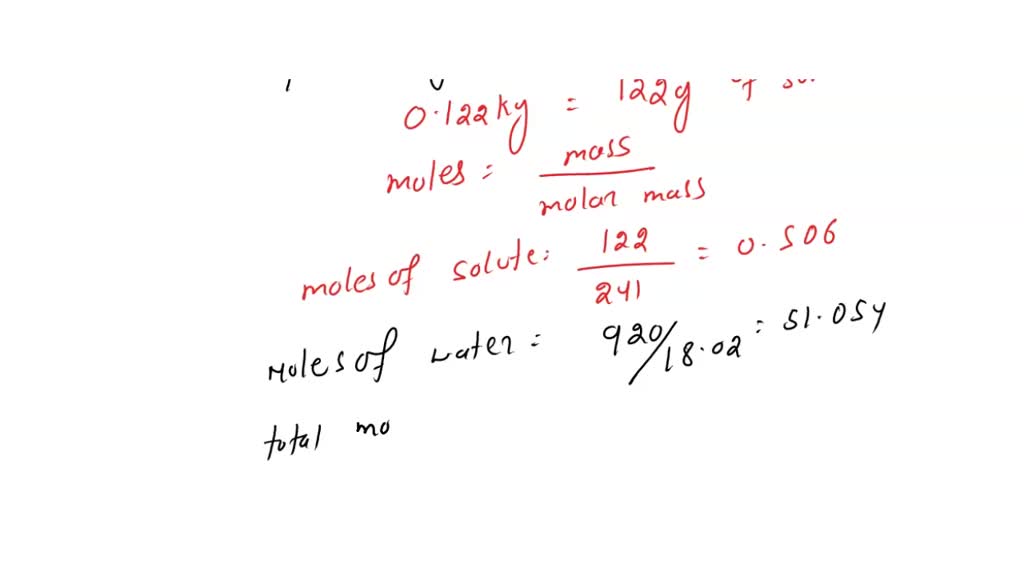
SOLVED: Given that p*(H2O) = 0.023 08 atm and p(H2O) = 0.022 39 atm in a solution in which 0.122 kg of a non-volatile solute (M = 241 g mol−1) is dissolved
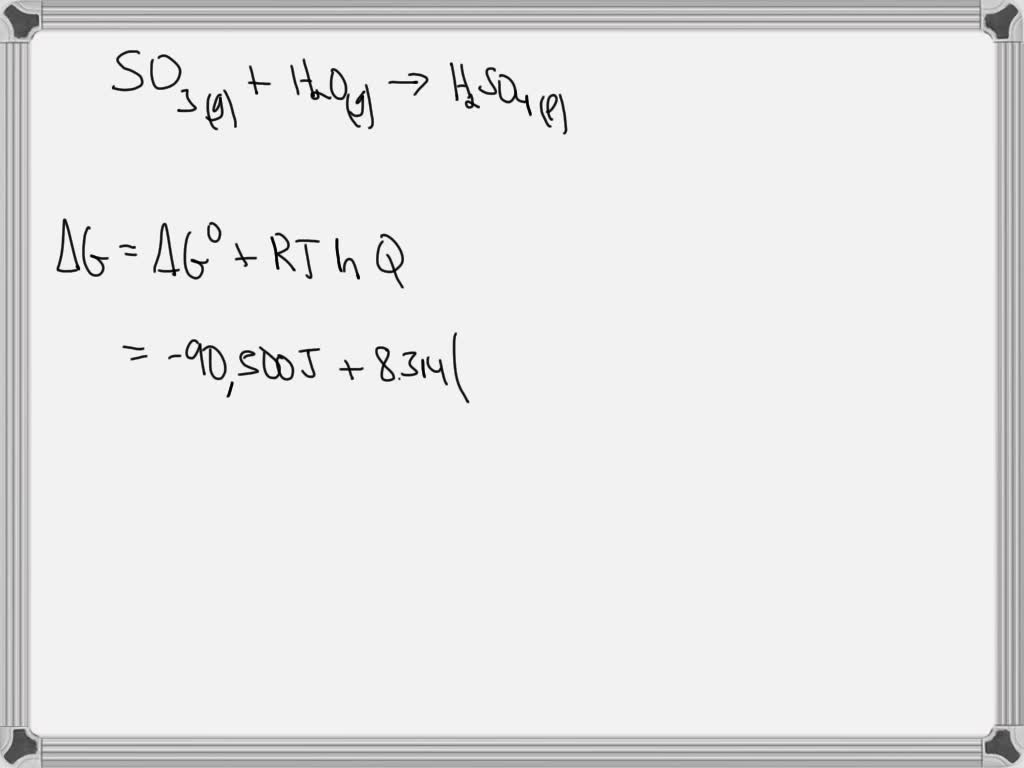
SOLVED: Calculate ΔGrxn (in kJ) at 298 K under the conditions shown below for the following reaction. SO3 (g) + H2O (g) → H2SO4 (l) ΔG°= -90.5 kJ P(SO3) = 0.20 atm, P(H2O) = 0.88 atm
9. Complete combustion of hydrocarbon P form x moles of H2O and y mol of CO2 .if x=1.5 y , the volume of CO2 produced at stp due to complete combustion of