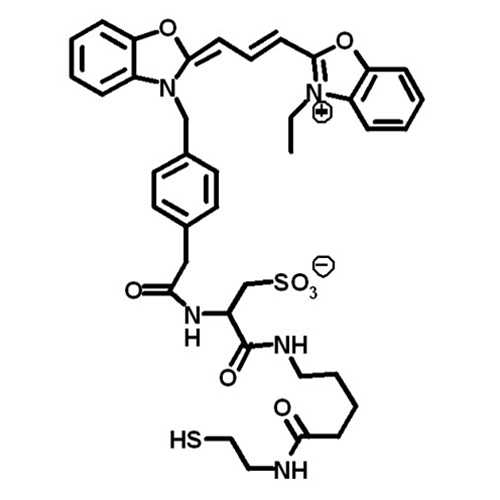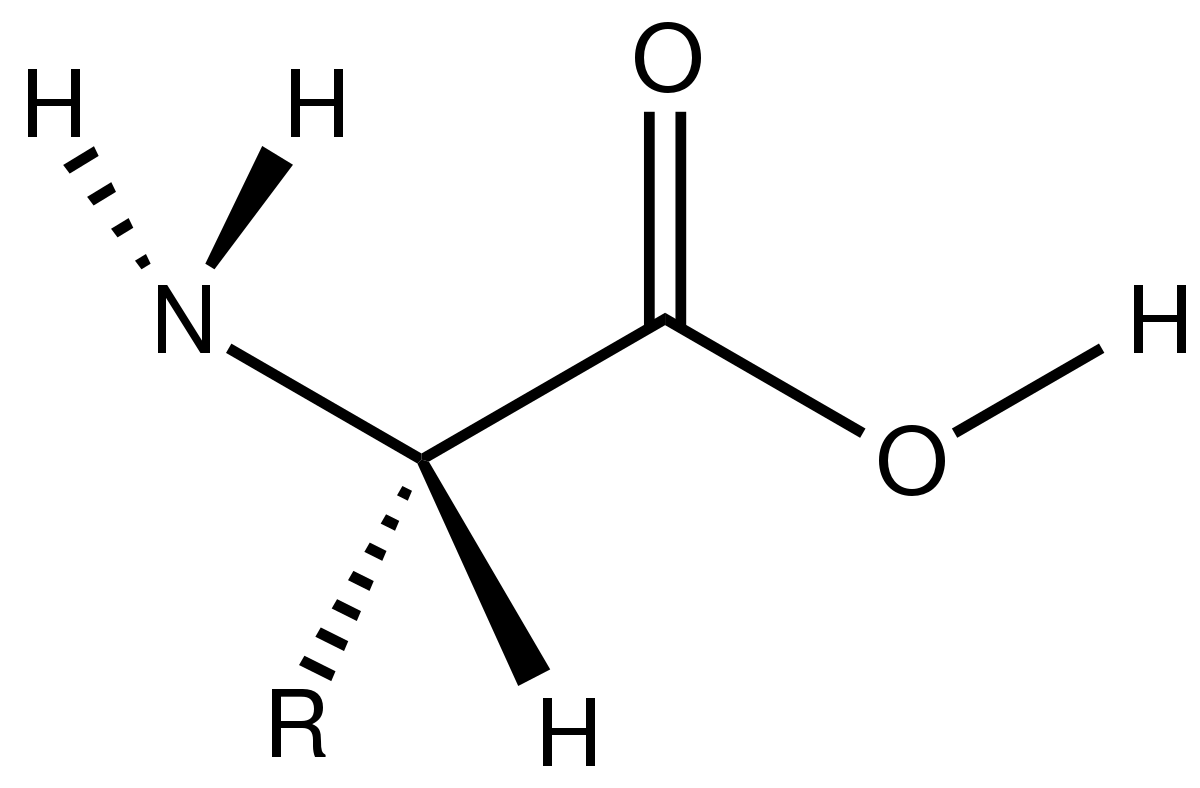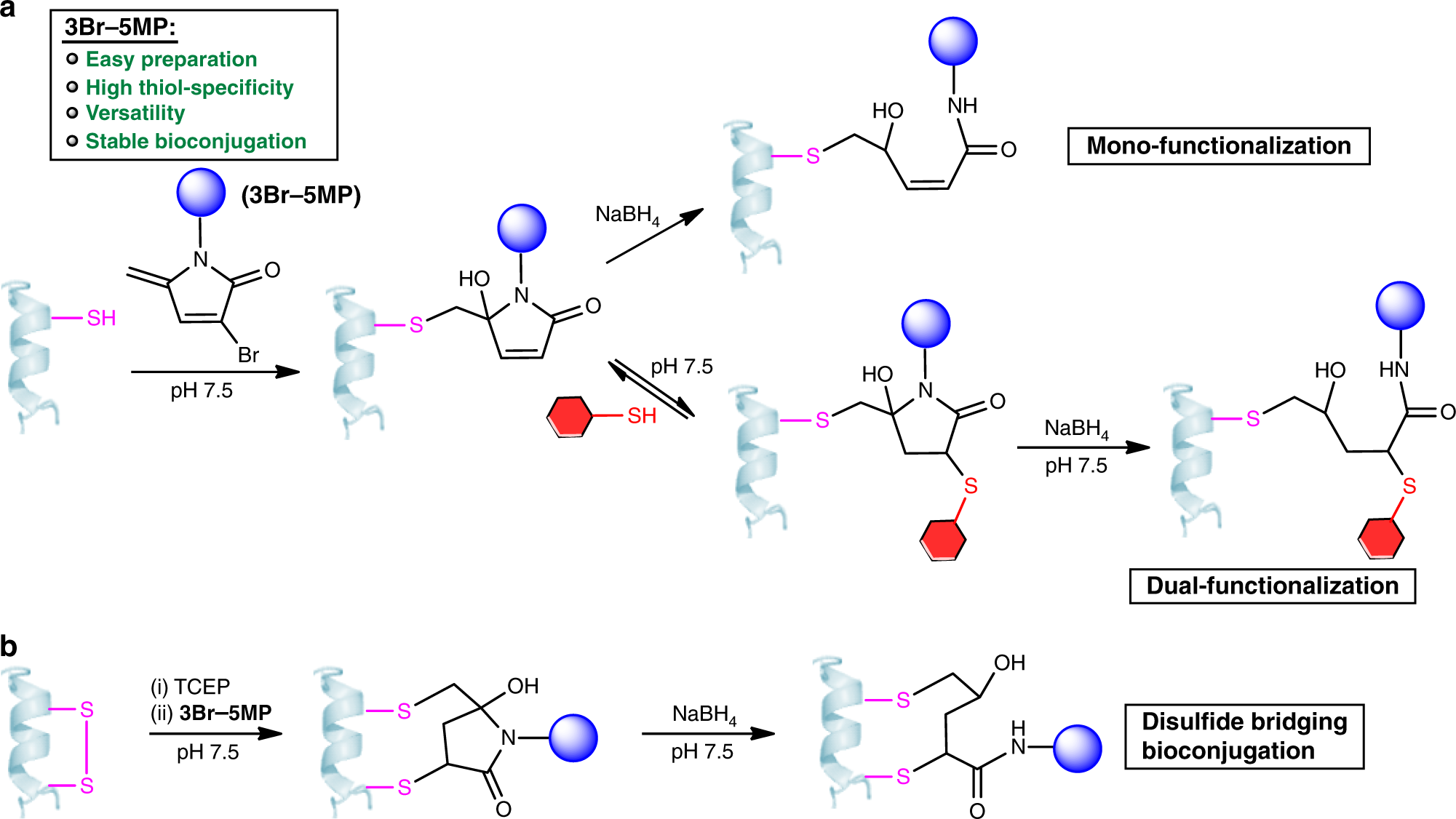
Cysteine-specific protein multi-functionalization and disulfide bridging using 3-bromo-5-methylene pyrrolones | Nature Communications
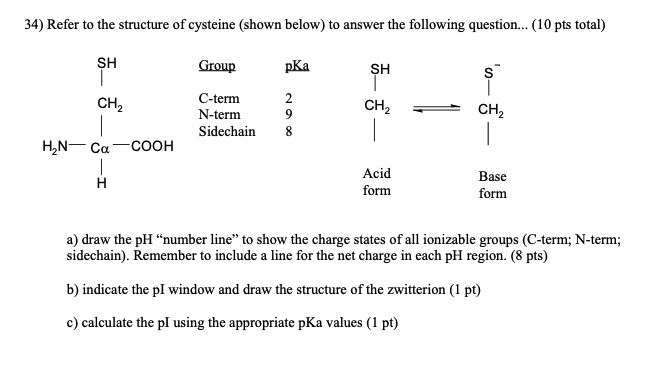
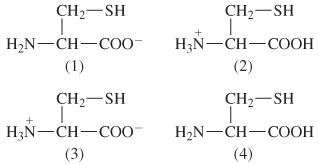
SOLVED: 34) Refer to the structure of cysteine (shown below) to answer the following question (10 pts total) SH Group pKa SH C-tern N-term Sidechain CHz CHz HzN Ca COOH Acid form

The Reactivity and Oxidation Pathway of Cysteine 232 in Recombinant Human α1-Antitrypsin - ScienceDirect

The pKa values for the amino acid cysteine are 1.8, 8.3, and 10.8. Assign these pKa values to the functional groups in cysteine and draw the structure of the molecule in aqueous
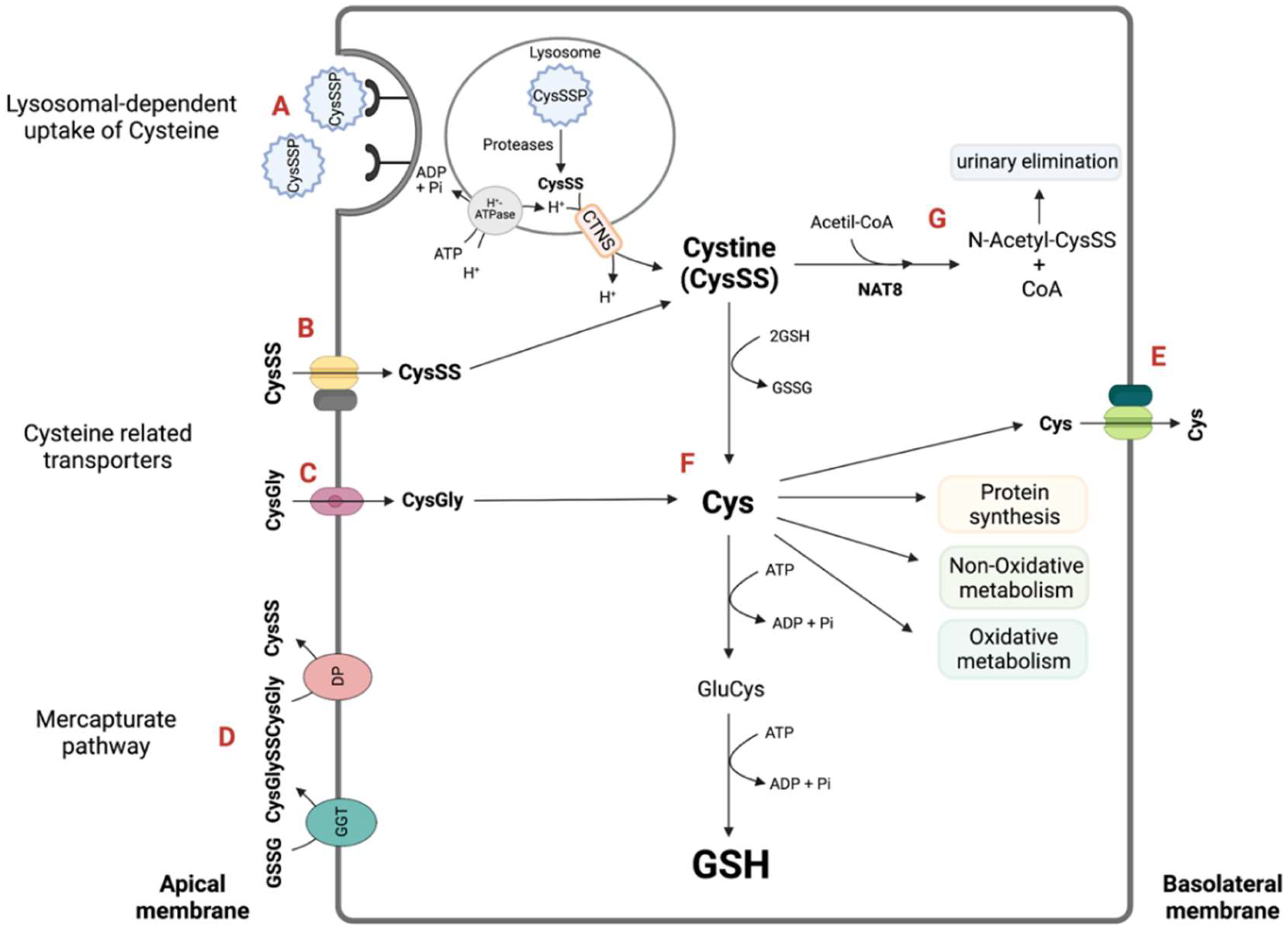
Molecules | Free Full-Text | Cysteine as a Multifaceted Player in Kidney, the Cysteine-Related Thiolome and Its Implications for Precision Medicine
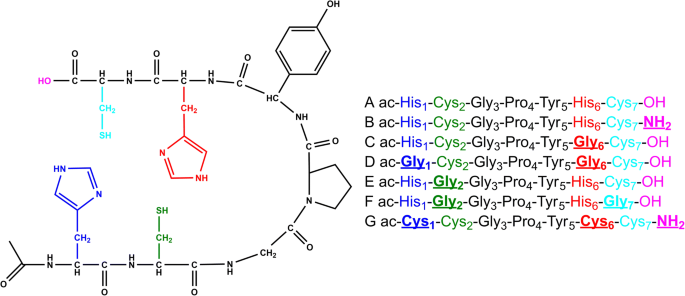
Weak Acid-Base Interactions of Histidine and Cysteine Affect the Charge States, Tertiary Structure, and Zn(II)-Binding of Heptapeptides | SpringerLink

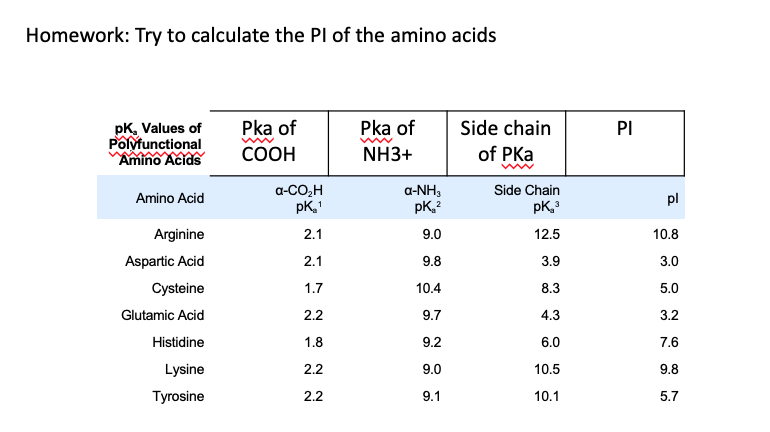
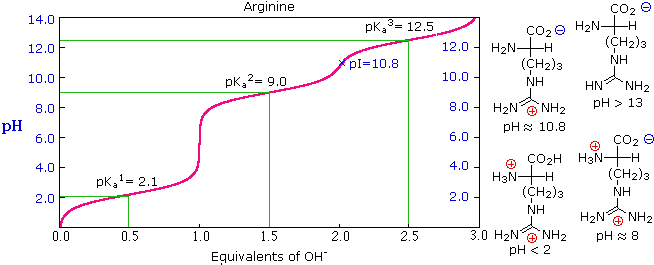
OneClass: Calculate the pI of cysteine. Use the pKa values given in Biochemical Calculations, appendi...
biochemistry - How do I calculate the isoelectric point of amino acids, each of which has more than two values of pKa? - Chemistry Stack Exchange