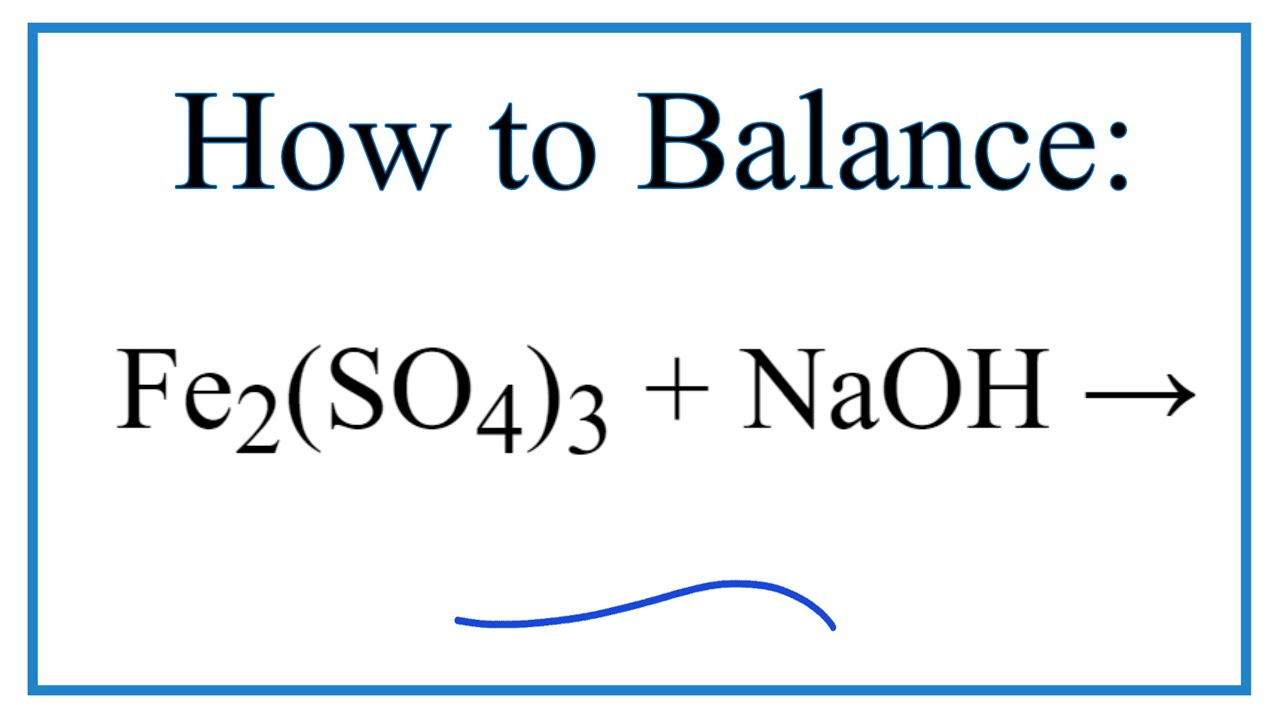
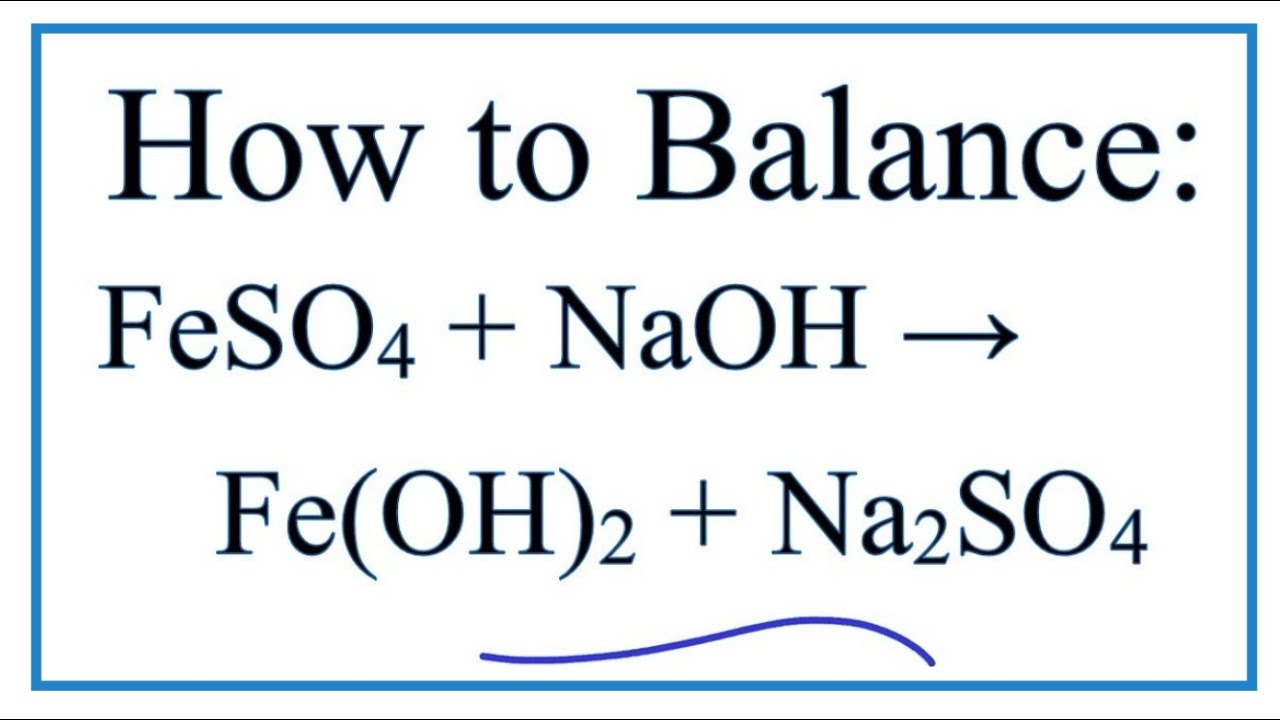
FeSO4+NaClO+NaOH=>Fe(OH)3+NaCl+Na2SO4+H2O Решить окислительно восстановительную реакцию - Школьные Знания.com

FeSO4 + 2 NaOH → Fe(OH)2 + Na2SO4 • The interaction between Fe (II) ions and sodium hydroxide. • Double Displacement reactions are a common occurrence on... | By The Cockney Chemist | Facebook

2.svg)