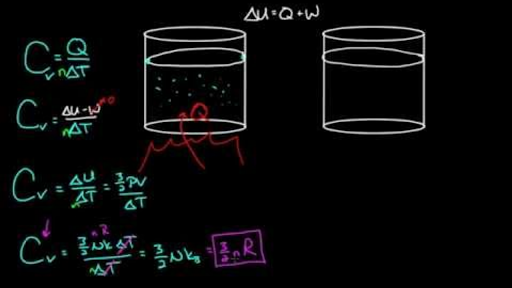
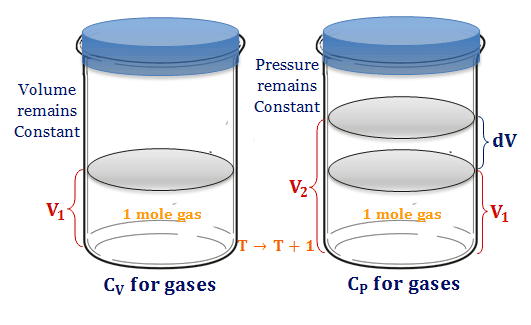
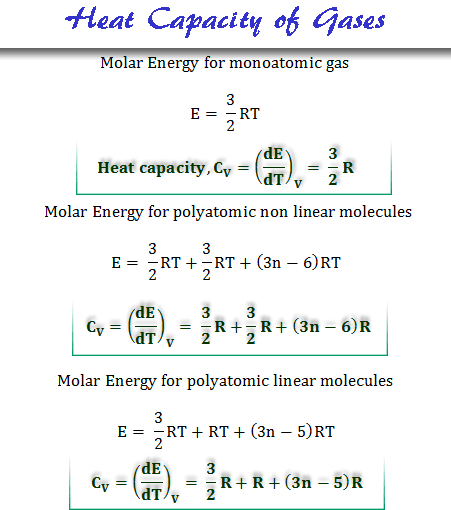Relation Between Heat capacity at Constant Volume (CV) & Constant Pressure (CP) - Chemistry - Aakash Byjus | AESL

Calculate the molar specific heat at constant volume . Given : specific heat of hydrogen at constant - YouTube

Calculate the specific heat capacity at constant volume for a gas. Given specific heat capacity at - YouTube

SOLVED: Question 3: The specific heat capacities at constant pressure and constant volume, Cpand Cv, are related by the following expression: Cp Cv = TV (@) where B = (7) , and

thermodynamics - Derivation of heat capacity at constant pressure and temperature - Physics Stack Exchange

The molar specific heat at constant volume of gas mixture is (13R)/(6). The gas mixture consists... - YouTube
The specific heat of a gas is found to be0.075 calories at constant volume and its formulawt is 40. The atomicity of the gas would be(1) One(2) Two(4) Four3) Three
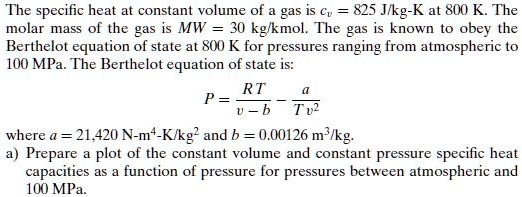
SOLVED: The specific heat constant volume of gas IS C = 825 Jlkg-K at 800 K The molar mass of the gas is MW = 30 kglkmol The g1s is known to