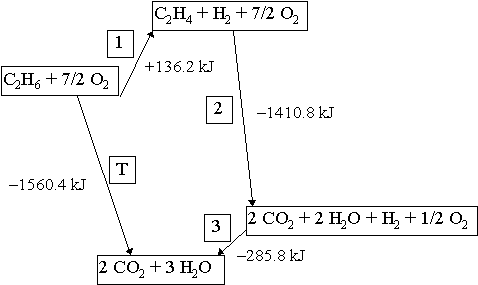
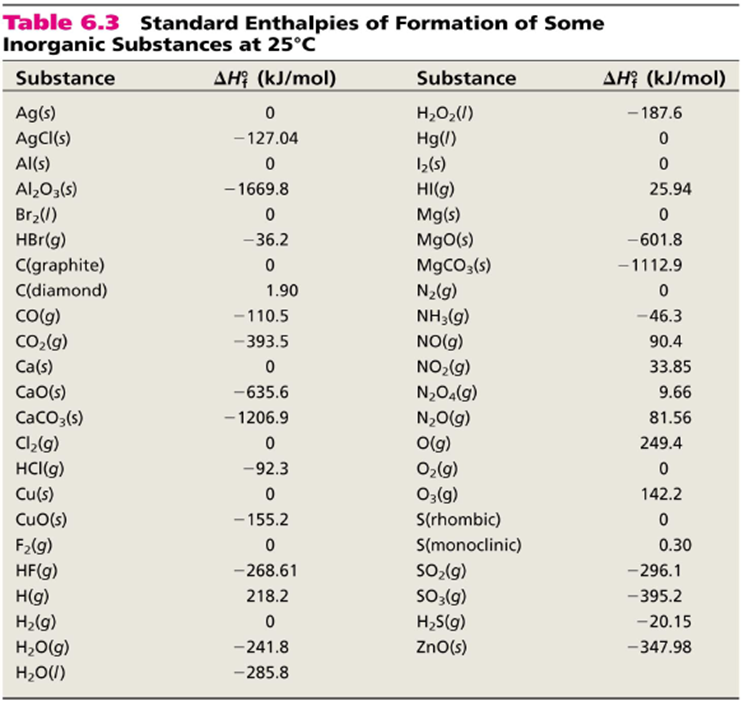
From the following reactions at 298 K .(A) CaC2(s) + 2H2O(l) → Ca(OH)2(s) + C2H2 (g); Δ H^∘ = - 127.9 kJ mol^-1 (B) Ca(s) + 12 O2(g) → CaO(s) ; Δ

Chapter 17 Thermochemistry. Thermochemistry: Study of energy changes that occur during chemical reactions and changes in state Section 17.1: The flow. - ppt download
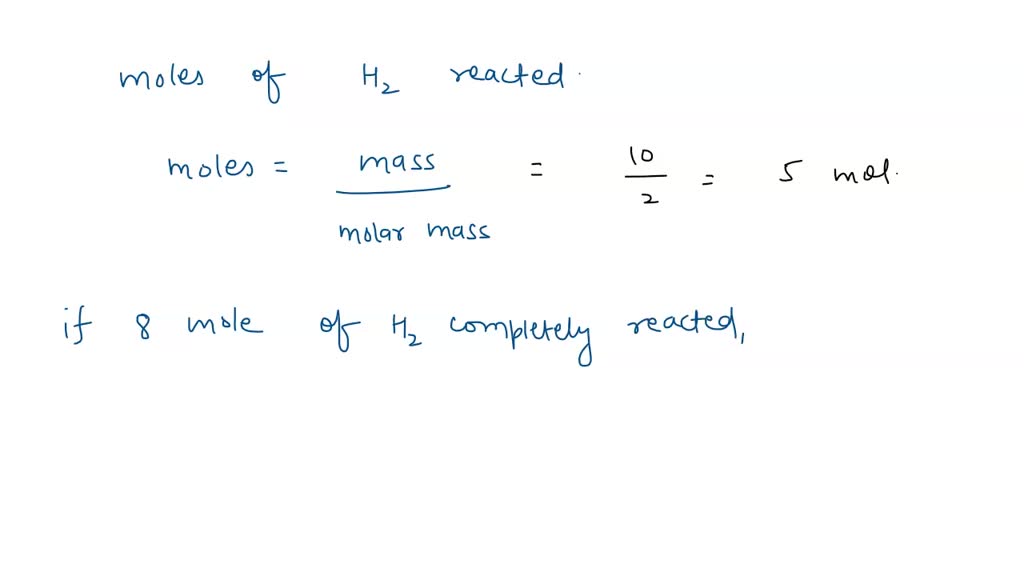
SOLVED: Consider the following thermochemical equation: S8 + 8 H2 → 8 H2S ∆rH = −161.6 kJ/mol-rxn a. Determine qrxn if 0.250 mol H2S is produced. b. Determine qrxn when 10.0 g