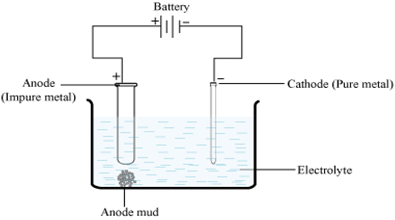
SOLVED: Among the metals, Ti, V, W, Zr, Th and Au, the no. of metals purified by van Arkel method is
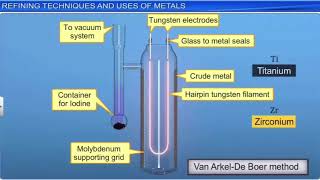
Describe the principle of van - arkel process for refining of metal. - Sarthaks eConnect | Largest Online Education Community

Answer the question number 14 a by an expert which i easely understand (4) (c) Calculate rate constant of the - Chemistry - Surface Chemistry - 13294039 | Meritnation.com

In van Arkel method, if `I_(2)` is introducted at `1800K` anode impure zironium metal, the product - YouTube
Describe principle of Van-Arkel Method methods in refining of metals. - Sarthaks eConnect | Largest Online Education Community
%2BMond%2BProcess%2Band%2B(2)%2B%2Bvan%2Barkel%2Bmethod.png)
Chemistry Notes Info - Your Chemistry Tutor provide notes for 9,10,11,12, BSc, MSc, Chemistry Quiz: 12 Class Chapter 6- General principle and process of isolation of the elements

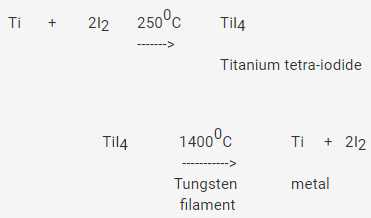
Write Chemical Reactions Involved in Van Arkel Method for Refining Titanium - Chemistry | Shaalaa.com

Two moles of an ideal gas at 2 bar and 27^∘C expand isothermally against a constant pressure of 1 bar. The work done by the gas is equal to :